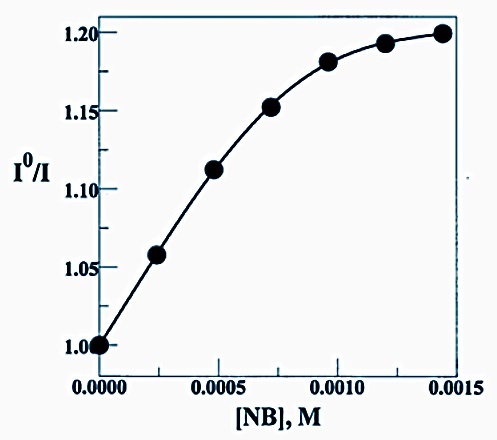
Fluorescence quenching of uric acid solubilized in bicontinuous microemulsion by nitrobenzene
Abstract
Full Text:
PDFReferences
- http://en.wikipedia.org/wiki/Uric_acid
- http://emedicine.medscape.com/article/244255-overview
- H.K. Choi, D.B. Mount, A.M. Reginato, Ann. Intern Med., 2005,143, 499-516
- A.E. Taylor, J. Biol. Chem., 1905, 1, 177-183
- M.E. Moran, Bioscience, 2003, 8, 1339-1355
- W. His, T. Paul, Zeitschr. f. physiol. Chem., 1900, xxxi, 1-42
- Y.Y. Sautin, T. Nakagawa, S. Zharikov, R. Johnson, Am. J. Physiol. – Cell Physiology, 2007, 293, C584-C596.
- C. Campos, R. Guzman, E. Lopez-Fernandez, A. Casado, Clinical Biochem., 2010, 43, 228-233.
- K.R. Daffner, J. Alzheimer’s Disease, 2010, 19, 1101-1122
- M. Boban, D. Modun, Croatia Me. J., 2010, 51 16-22
- B.F. Becker, N. Reinholz, B. Leipert, P. Raschke, B. Permanetter, E. Gerlach, Chest, 1991, 100, 176S-181S
- F.J. Nieto, C. Iribarren, M.D. Gross, G.W. Comstock, R.G. Cutler, Atherosclerosis, 2000, 148, 131-139
- M. Godycki-Cwrko, M. Krol, A. Zwolinska, K. Kolodziejczyk, M. Kasielski, G. Padula, J. Grebocki, P. Kazimierska, M. Miatkowski, J. Markowski, D. Nowak, J. Am. Coll. Nutrition, 2010, 29, 397-406
- W.S. Waring, QJ Med., 2002, 95, 691-693
- P.H. Proctor, Stroke, 2008, 39, e126
- B.N. Ames, R. Cathcart, E. Schwiers, P. Hochstein, Proc. Natl. Acad. Sci. USA, 1981, 78, 6858-6862
- G.G. Warr, R. Sen, D.F. Evans, J.E. Trend, J. Phys. Chem., 1988, 92,774-783
- D.F. Evans, D.J. Mitchell, B.W. Ninham, Ibid, 1986, 90, 2817-2825
- K. Shinoda, B. Lindman, Langmuir, 1987, 3, 135-149
- L.R. Angel, D.F. Evans, B.W. Ninham, J. Phys. Chem., 1983, 87, 538-540
- M.O. Iwunze, A. Sucheta, J.F. Rusling, Anal. Chem., 1990, 62, 644-649
- R. Johannsson, M. Almgren, R. Schomacker, Langmuir, 1993, 9, 1269-1273
- R. Rusakowicz, A.C. Testa, Ibid, 1968, 72, 793-796
- J.N. Demas, G.A. Crosby, J. Phys. Chem., 1971, 75, 991-1024
- S.J. Strickler, R.A. Berg, J. Chem. Phys., 1962, 37, 814-822
- http://www.wolframalpha.com/entities/chemicals/uric_acid/fn/pf/pq
DOI: http://dx.doi.org/10.13171/mjc.2.3.2013.28.02.08
Refbacks
- There are currently no refbacks.
Copyright (c) 2015 Mediterranean Journal of Chemistry
