DFT-based QSAR studies and Molecular Docking of 1-Phenylcyclohexylamine Analogues as anticonvulsant of NMDA Receptor
Abstract
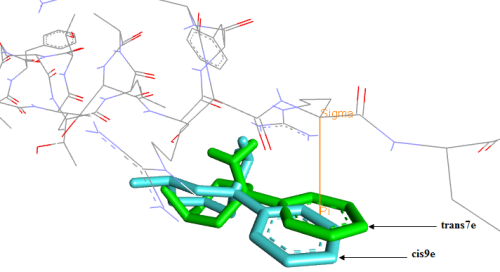
The phencyclidine (PCP) and their analogues have been reported to exhibit inhibitory activities toward the N-methyl-D-aspartate receptor (NMDAR). To discover the QSAR between structure of PCP derivatives and Ki activities we have used density functional theory (DFT) to generate quantum descriptors, multiple regression linear (MLR) method was applied to establish QSAR model, and an artificial neural network (ANN), considering the relevant descriptors obtained with the MLR method is explored, a correlation coefficient of RANN = 0.912 was obtained with 6-4-1 ANN model. This model is tested by using a cross-validation method with the LOO procedure (RCV = 0.841). To study the configuration impact on activity, we proceed to the Molecular Docking of four configurations, two configurations of compound have (Ki = 502 nM) and two configurations of compound have (Ki = 1200nM). The phenyl group, when placed in an equatorial position in cis9e, a configuration of the less active compound, does not form π-sigma interaction. The superimposition of this configuration with trans7e reveals that the phenyl group of cis9e configuration is shifted from the binding site compared to trans7e which forms an interaction π-sigma throughout its phenyl group with ARG B: 894. So, we could claim that the cis9e is the configuration adopted by compound having (Ki = 502 nM).
Full Text:
PDFReferences
- R.G. Morris, E. Anderson, G.S. Lynch, M. Baudry, “Selective impairment of learning and blockade of long-term potentiation by an N-methyl-d-aspartate receptor antagonist, AP5”, Nature, 319, 1986, 774–776.
- T. Kutsuwada, N. Kashiwabuchi, H. Mori, K. Sakimura, E. Kushiya, K. Araki, H. Meguro, H. Masaki, T. Kumanishi, M. Arakawa,”Molecular diversity of the NMDA receptor channel”, Nature, 358, 1992, 36–41.
- K. Nakazawa, M.C. Quirk, R.A. Chitwood, M. Watanabe, M.F. Yeckel, L.D. Sun, A. Kato, C.A. Carr, D. Johnston, M.A. Wilson, S. Tonegawa, “Requirement for hippocampal CA3 NMDA receptors in associative memory recall”, Science, 297, 2002, 211–218.
- A. Sanz-Clemente, R.A. Nicoll, K.W. Roche, “Diversity in NMDA receptor composition: many regulators, many consequences”, Neuroscientist, 19, 2013, 62−75.
- P. Paoletti, C. Bellone, Q. Zhou, “NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease”, Nature Reviews Neuroscience, 14, 2013, 383−400.
- S.F. Traynelis, L.P. Wollmuth, C.J. McBain, F.S. Menniti, K.M. Vans, K.K. Ogden, K.B. Hansen, H. Yuan, S.J. Myers, R. Dingledine, “Glutamate receptor ion channels: structure, regulation, and function”, Pharmacological Reviews, 62, 2010, 405–496.
- H. Monyer, R. Sprengel, R. Schoepfer, A. Herb, M. Higuchi, H. Lomeli, N. Burnashev, B. Sakmann, PH. Seeburg, “Heteromeric NMDA receptors: molecular and functional distinction of subtype”, Science, 256, 1992, 1217–1221.
- R.A. Al-Hallaq, T.P. Conrads, T.D. Veenstra, R.J. Wenthold, “NMDA di-heteromeric receptor populations and associated proteins in rat hippocampus”, Journal of Neuroscience, 27, 2007, 8334−8343.
- S.L.C. Brothwell, J.L. Barber, D.T. Monaghan, D.E. Jane, A.J. Gibb, S. Jones, “NR2B- and NR2D-containing synaptic NMDA receptors in developing rat substantia nigra pars compacta dopaminergic neurons”, Journal of Physiology, 586, 2008, 739−750.
- M.A. Rogawski, “therapeutic potential of excitatory amino acid antagonists: channel blockers and 2,3-benzodiazepines”, Trends in Pharmacological Sciences, 14, 1993, 325-331.
- P. D. Leeson, L. L. Iversen, “The glycine site on the NMDA receptor: Structure-Activity relationships and therapeutic potential”, Journal of Medicinal Chemistry, 37, 1994, 4053-4067.
- G.L. Collingridge, A. Volianskis, N. Bannister, G. France, L. Hanna, M. Mercier, P. Tidball, G. Fang, M.W. Irvine, B.M. Costa, D.T. Monaghan, Z.A. Bortolotto, E. Molnar, D. Lodge, D.E. Jan, “The NMDA receptor as a target for cognitive enhancement”, Neuropharmacology, 64, 2013, 13-26.
- B.M. Costa, M.W. Irvine, G. Fang, R.J. Eaves, M.B. Mayo-Martin, B. Laube, D.E. Jane, D.T. Monaghan, “Structure-activity relationships for allosteric NMDA receptor inhibitors based on 2-naphthoic acid”, Neuropharmacology, 62, 2012, 1730-1736.
- B.M. Costa, M.W. Irvine, R.J. Eaves, M.B. Mayo-Martin, D.A. Skifter, D.E. Jane, D.T. Monaghan, “A novel family of negative and positive allosteric modulators of NMDA receptors”, Journal of Pharmacology and Experimental Therapeutics, 335(3), 2010, 614-621.
- M.W. Irvine, B.M. Costa, A. Volianskis, G. Fang, L. Ceolin, G.L. Collingridge, D.T. Monaghan, D.E. Jane, “Coumarin-3-carboxylic acid derivatives as potentiators and inhibitors of recombinant and native N-Methyl-D-aspartate receptors”, Neurochemistry International, 61, 2012, 593-600.
- D.T. Monaghan, M.W. Irvine, B.M. Costa, G. Fang, D.E. Jane, “Pharmacological modulation of NMDA receptor activity and the advent of negative and positive allosteric modulators”, Neurochemistry International, 61(4), 2012, 581-592.
- M.W. Irvine, B.M. Costa, D. Dlaboga, G. Culley, R. Hulse, C.L. Scholefield, P. Atlason, G. Fang, R. Evals, R. Morley, M.B. Mayo-Martin, M. Amici, Z.A. Bortolotto, L. Donaldson, G.L. Collingridge, E. Molnar, D.T. Monaghan, D.E. Jane, “Piperazine-2,3-dicarboxylic acid derivatives as dual antagonists of NMDA and GluK1-containing kainate receptors”, Journal of Medicinal Chemistry, 55, 2012, 327-341.
- M. Elhallaoui, M. Laguerre, A. Carpy, O.F. Chahdi, “Molecular Modeling of non-competitive antagonists of the NMDA receptor: proposal of pharmacophore and a description of interaction model”, Journal of Molecular Modeling, 8, 2002, 65-72.
- A. Thurkauf, B. Costa, S. Yamaguchi, M.V. Mattson, A.E. Jacobson, K.C. Rice, M.A. Rogawski, “Synthesis and Anticonvulsant Activity of 1-Phencyclohexylamine Analogues”, Journal of Medicinal chemistry, 33, 1990, 5.
- C.T. Lee, W.T. Yang, R.G. Parr, “Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density”, Physical Review B, 37, 1988, 785-789.
- E. Niecke, P. Becker, M. Nieger, D. Stalke, W. W. Schoeller, “Carbenoid or Lithium Complex of a carbanion? Synthesis and Structure of (Me3Si)2C=P(aryl)=C(Cl)Li(thf)3 and LiCl Elimination To Give the Phosphiren”, Chemie International Edition in English, 34, 1995, 1849-1852.
- M.J. Frish, G. W. Turcks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. J. A. Montgomery, T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Lyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y. Nakatsuji, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, Revision D.01, Gaussian, Inc., Wallingford, CT, 2004
- Advanced Chemistry Development Inc., Toronto, Canada (2009). (www.acdlabs.com/resources/freeware/chemsketch/).
- C. El M’barki, M. Elhallaoui, “Quantum Calculations to Construct a 3D-QSAR Model Based on PCP-TCP Derivatives and Molecular Docking with NMDA Receptor”, Journal of Materials and Environmental Science,8 (4), 2017,1391-1400.
- H. Hadni, M. Mazigh, E. Charif, A. Bouayad, M. Elhallaoui, Molecular Modeling of Antimalarial Agents by 3D-QSAR Study and Molecular Docking of Two Hybrids 4-Aminoquinoline-1,3,5-triazine and 4-Aminoquinoline-oxalamide Derivatives with the Receptor Protein in Its Both Wild and Mutant Types, Biochem. Res. Int., 2018, 1–15.
- AutoDockTools, version 1.5.6 Sep_17_14.
- G. M. Morris, R. Huey, W. Lindstrom, M. F. Sanner, R. K. Belew, D. S. Goodsell, A. J. Olson,” Autodock4 and AutoDockTools4: automated docking with selective receptor flexibility”, Journal of Computational Chemistry, 16, 2009, 2785–2791.
- Z. Akyol, L. Gakhar, B. R. Sorensen, J. H. Hell, M. A. Shea, “The NMDA Receptor NR1 C1 Region Bound to Calmodulin: Structural Insights into Functional Differences between Homologous Domains”, Structure, 15, 2007, 1603-1617.
- Discovery Studio Visualizer v4.1.0.14169 Copyright © 2005-14, Accelrys Software Inc.
- H. Hadni, M. Mazigh, E. Charif, A. Bouayad, M. Elhallaoui, Molecular Modeling of Antimalarial Agents by 3D-QSAR Study and Molecular Docking of Two Hybrids 4-Aminoquinoline-1,3,5-triazine and 4-Aminoquinoline-oxalamide Derivatives with the Receptor Protein in Its Both Wild and Mutant Types, Biochem. Res. Int., 2018, 1–15.
- S. S. So, W. G. Richards, “Application of Neural Networks: Quantitative Structure-Activity Relationships of the Derivatives of 2,4-Diamino-5- (substituted-benzyl) pyrimidines as DHFR Inhibitors”, Journal of Medicinal Chemistry, 35, 1992, 3201-3207.
- D. Zakarya, D. Cherqaoui, M. Esseffar, D. Villemin, J. M. Cense, “Application of Neural Networks to Structure-Sandalwood Odour Relationships”, Journal of Physical Organic Chemistry, 10, 1997, 612-622.
DOI: http://dx.doi.org/10.13171/mjc01912121044hh
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
