Electrochemical oxidation of textile azo dye reactive orange 16 on the Platinum electrode
Abstract
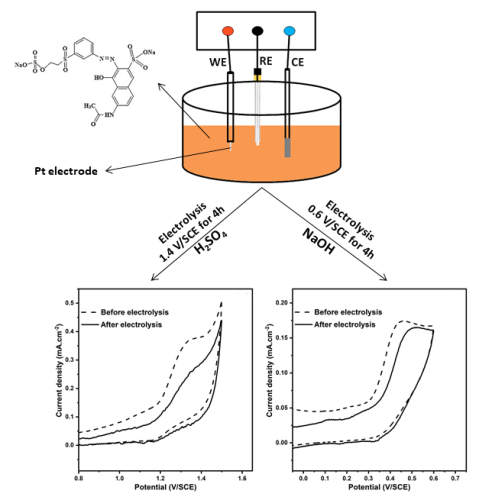
This study focused mainly on the color removal of textile azo dye Reactive Orange 16 (RO16) by electrochemical oxidation. The effect of supporting electrolyte (H2SO4 and NaOH), RO16 concentration (from 0.5 to 10 mM) and potential scan rate (between 20 and 500 mV/s) was performed with cyclic voltammetry using platinum (Pt) wire as working electrode. The anodic peak current density was linear to RO16 concentrations. This allows the lowest concentrations to be determined voltammetrically in the two electrolytic media, acid (H2SO4 1 M) and alkaline (NaOH 0.1 M). Linearity between the current density and the square root of the potential scan rate was observed in both electrolytes. This means that the electrochemical reaction at the electrode-electrolyte interface is controlled by the diffusion process. The slope of the logarithm of peak current density versus the logarithm of potential scan rate was found to be 0.43 for RO16 in H2SO4 and 0.48 in NaOH these values of slop are close to the thеoretical value of 0.5 which confirms the diffusion process. The removal efficiency of the dye in acid electrolyte reached 40%, while it is 18% in the basic media after 4 hours of electrolysis by chronoamperometry.
Full Text:
PDFReferences
- A. Gürses, M. Açıkyıldız, K. Güneş, M. S. Gürses, Dyes and Pigments: Their Structure and Properties, 2016, pp. 13–29.
- V. J. P. Poots, G. McKay, J. J. Healy, The removal of acid dye from effluent using natural adsorbents-I peat, Water Res., 1976, 10,
–1066.
- A. A. Kadam, A. A. Telke, S. S. Jagtap, S. P. Govindwar, Decolorization of adsorbed textile dyes by a developed consortium of Pseudomonas sp. SUK1 and Aspergillus ochraceus NCIM-1146 under solid-state fermentation, J. Hazard. Mater., 2011, 189, 486–494.
- A. Hasanbeigi, L. Price, A technical review of emerging technologies for energy and water efficiency and pollution reduction in the textile industry, J. Clean. Prod., 2015, 95, 30–44.
- H. Y. El-Kassas, L. A. Mohamed, Bioremediation of the textile waste effluent by Chlorella vulgaris, Egypt. J. Aquat. Res., 2014, 40, 301–308.
- O. Yesilada, D. Asma, S. Cing, Decolorization of textile dyes by fungal pellets, Process Biochem., 2003, 38, 933–938.
- I. M. Banat, P. Nigam, D. Singh, R. Marchant, Microbial decolorization of textile-dyecontaining effluents: A review, Bioresour. Technol., 1996, 58, 217–227.
- T. Cheunbarn, S. Cheunbarn, T. Khumjai, Prospects of bacterial granule for treatment of real textile industrial wastewater, Int J Agric Biol., 2008, 10, 689–92.
- N. Muhd Julkapli, S. Bagheri, S. Bee Abd Hamid, Recent advances in heterogeneous photocatalytic decolorization of synthetic dyes, Sci. World J., 2014, 2014, 25.
- V. K. Sharma, Aggregation and toxicity of titanium dioxide nanoparticles in aquatic environment-A Review, J. Environ. Sci. Heal. Part A., 2009, 44, 1485–1495.
- H. S. Lade, T. R. Waghmode, A. A. Kadam, S. P. Govindwar, Enhanced biodegradation and detoxification of disperse azo dye Rubine GFL and textile industry effluent by the defined fungal-bacterial consortium, Int. Biodeterior. Biodegradation., 2012, 72, 94–107.
- S. Dixit, A. Yadav, P. D. Dwivedi, M. Das, Toxic hazards of leather industry and technologies to combat threat: A review, J. Clean. Prod., 2015, 87, 39–49.
- M. -M. Zhang, W. -M. Chen, B. -Y. Chen, C. T. Chang, C. -C. Hsueh, Y. Ding, K. -L. Lin, H. Xu, Comparative study on characteristics of azo dye decolorization by indigenous decolorizers, Bioresour. Technol., 2010, 101, 2651–2656.
- F. P. Van der Zee, F. J. Cervantes, Impact and application of electron shuttles on the redox (bio)transformation of contaminants: A review, Biotechnol. Adv., 2009, 27, 256–277.
- R. G. Saratale, G. D. Saratale, J. S. Chang, S. P. Govindwar, Ecofriendly degradation of sulfonated diazo dye C.I. Reactive Green 19A using Micrococcus glutamicus NCIM-2168, Bioresour. Technol., 2009, 100, 3897–3905.
- H. Ben Mansour, D. Barillier, D. Corroler, K. Ghedira, L. Chekir-Ghedira, R. Mosrati, In Vitro Study Of DNA Damage Induced By Acid Orange 52 and its Biodegradation Derivatives, Environ. Toxicol. Chem., 2009, 28, 489.
- C. G. Boer, L. Obici, C. G. M. de Souza, R. M. Peralta, Decolorization of synthetic dyes by solid-state cultures of Lentinula (Lentinus) edodes producing manganese peroxidase as the main ligninolytic enzyme, Bioresour. Technol., 2004, 94, 107–112.
- C. Özdemir, M. K. Öden, S. Şahinkaya, E. Kalipçi, Color Removal from Synthetic Textile Wastewater by Sono-Fenton Process, CLEAN- Soil, Air, Water., 2011, 39, 60–67.
- C. A. Martínez-Huitle, E. Brillas, Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: A general review, Appl. Catal. B Environ., 2009, 87, 105–145.
- J. M. Peralta-Hernández, M. Méndez-Tovar, R. Guerra-Sánchez, C. A. Martínez-Huitle, J. L. Nava, A Brief Review on Environmental Application of Boron Doped Diamond Electrodes as a New Way for Electrochemical Incineration of Synthetic Dyes, Int. J. Electrochem., 2012, 2012, 1–18.
- S. M. de A. G. U. de Souza, K. A. S. Bonilla, A. A. U. de Souza, Removal of COD and color from hydrolyzed textile azo dye by combined ozonation and biological treatment, J. Hazard. Mater., 2010, 179, 35–42.
- E. R. Bandala, M. A. Peláez, A. J. García-López, M. de J. Salgado, G. Moeller, Photocatalytic decolourisation of synthetic and real textile wastewater containing benzidine-based azo dyes, Chem. Eng. Process. Process Intensif., 2008, 47, 169–176.
- A. A. Pourbabaee, F. Malekzadeh, M. N. Sarbolouki, F. Najafi, Aerobic Decolorization and Detoxification of a Disperse Dye in Textile Effluent by a New Isolate of Bacillus sp., Biotechnol. Bioeng., 2006, 93, 631–635.
- E. Forgacs, T. Cserháti, G. Oros, Removal of synthetic dyes from wastewaters: a review, Environ. Int., 2004, 30, 953–971.
- T. Robinson, G. McMullan, R. Marchant,
P. Nigam, Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative, Bioresour. Technol., 2001, 77, 247–255.
- F. Moughaoui, A. Ouaket, A. Laaraibi, S. Hamdouch, Z. Anbaoui, A. Abourriche, M. Berrada, A novel approach for producing low cost and highly efficient activated carbon for removing cationic dyes, Mediterr. J. Chem., 2019, 8, 74–83.
- Z. Zakaria, M. R. Othman, S. Z. Hasan, W. Y. Wan Ahmad, Electrochemical Degradation of Reactive Orange 16 by using Charcoal-Based Metallic Composite Electrodes, Sains Malaysiana., 2019, 48, 791–801.
- A. AM, F. Ouzidan, T. Ainane, M. Talbi, M. El Kouali, Adsorption of cationic dye onto Moroccan natural rock, Mediterr. J. Chem., 2019, 9, 37.
- B. Mella, B. S. de C. Barcellos, D. E. da Silva Costa, M. Gutterres, Treatment of Leather Dyeing Wastewater with Associated Process of Coagulation-Flocculation/Adsorption/Ozonation, Ozone Sci. Eng., 2018, 40, 133–140.
- K. Paździor, A. Klepacz-Smółka, S. Ledakowicz, J. Sójka-Ledakowicz, Z. Mrozińska, R. Żyłła, Integration of nanofiltration and biological degradation of textile wastewater containing azo dye, Chemosphere., 2009, 75, 250–255.
- J. P. Jadhav, G. K. Parshetti, S. D. Kalme, S. P. Govindwar, Decolourization of azo dye methyl red by Saccharomyces cerevisiae MTCC 463, Chemosphere., 2007, 68, 394–400.
- M. Behnajady, N. Modirshahla, H. Fathi, Kinetics of decolorization of an azo dye in UV alone and UV/H2O2 processes, J. Hazard. Mater., 2006, 136, 816–821.
- V. Erady, R. J. Mascarenhas, A. K. Satpati, A. K. Bhakta, Z. Mekhalif, J. Delhalle, A. Dhason, Carbon paste modified with Bi decorated multi-walled carbon nanotubes and CTAB as a sensitive voltammetric sensor for the detection of Caffeic acid, Microchem. J., 2019, 146,
–82.
- N. P. Shetti, D. S. Nayak, S. J. Malode, R. M. Kulkarni, D. B. Kulkarni, R. A. Teggi, V. V. Joshi, Electrooxidation and determination of flufenamic acid at graphene oxide modified carbon electrode, Surfaces and Interfaces., 2017, 9, 107–113.
- S. D. Bukkitgar, N. P. Shetti, Electrochemical behavior of an anticancer drug 5-fluorouracil at methylene blue modified carbon paste electrode, Mater. Sci. Eng. C., 2016, 65, 262–268.
- N. P. Shetti, D. S. Nayak, G. T. Kuchinad, R. R. Naik, Electrochemical behavior of thiosalicylic acid at γ-Fe2O3 nanoparticles and clay composite carbon electrode, Electrochim. Acta., 2018, 269, 204–211.
- U. Ghimire, M. Jang, S. Jung, D. Park, S. Park, H. Yu, S. -E. Oh, Electrochemical Removal of Ammonium Nitrogen and COD of Domestic Wastewater using Platinum Coated Titanium as an Anode Electrode, Energies, 2019, 12, 883.
- M. Jović, D. Stanković, D. Manojlović, I. Anđelković, A. Milić, B. Dojčinović, G. Roglić, Study of the Electrochemical Oxidation of Reactive Textile Dyes Using Platinum Electrode, Int. J. Electrochem. Sci., 2013, 8, 168–183.
- O. J. D’Souza, R. J. Mascarenhas, A. K. Satpati, B. M. Basavaraja, A novel ZnO/reduced graphene oxide and Prussian blue modified carbon paste electrode for the sensitive determination of Rutin, Sci. China Chem., 2019, 62, 262–270.
- D. B. Shikandar, N. P. Shetti, R. M. Kulkarni, S. D. Kulkarni, Silver-doped titania modified carbon electrode for electrochemical studies of furantril, ECS J. Solid State Sci. Technol., 2018, 7, Q3215–Q3220.
- S. Kumar, S. D. Bukkitgar, S. Singh, Pratibha, V. Singh, K. R. Reddy, N. P. Shetti, C. Venkata Reddy, V. Sadhu, S. Naveen, Electrochemical Sensors and Biosensors Based on Graphene Functionalized with Metal Oxide Nanostructures for Healthcare Applications, ChemistrySelect., 2019, 4, 5322–5337.
- A. K. Bhakta, S. Kumari, S. Hussain, P. Martis, R. J. Mascarenhas, J. Delhalle, Z. Mekhalif, Synthesis and characterization of maghemite nanocrystals decorated multi-wall carbon nanotubes for methylene blue dye removal, J. Mater. Sci., 2019, 54, 200–216.
- N. Shetti, D. Nayak, Electrochemical detection of chlorpheniramine maleate in the presence of an anionic surfactant and its analytical applications, Can. J. Chem., 2017, 95, 553–559.
- S. D. Bukkitgar, N. P. Shetti, R. M. Kulkarni, K. R. Reddy, S. S. Shukla, V. S. Saji, T. M. Aminabhavi, Electro-catalytic behavior of Mg-doped ZnO nano-flakes for oxidation of anti-inflammatory drug, J. Electrochem. Soc., 2019, 166, B3072–B3078.
- N. P. Shetti, S. J. Malode, R. S. Malladi, S. L. Nargund, S. S. Shukla, T. M. Aminabhavi, Electrochemical detection and degradation of textile dye Congo red at graphene oxide modified electrode, Microchem. J., 2019, 146, 387–392.
- C. -Y. Chen, Photocatalytic Degradation of Azo Dye Reactive Orange 16 by TiO2, Water. Air. Soil Pollut., 2009, 202, 335–342.
- L. S. Andrade, T. T. Tasso, D. L. da Silva, R. C. Rocha-Filho, N. Bocchi, S. R. Biaggio, On the performances of lead dioxide and boron-doped diamond electrodes in the anodic oxidation of simulated wastewater containing the Reactive Orange 16 dye, Electrochim. Acta., 2009, 54, 2024–2030.
- S. W. Won, S. B. Choi, Y. S. Yun, Performance and mechanism in binding of Reactive Orange 16 to various types of sludge, Biochem. Eng. J., 2006, 28, 208–214.
- Z. Zakaria, N. Nordin, S. Zubaidah Hasan, N. Afzalina Baharuddin, M. Ahmed Jumaah, M. Rozali Othman, Decolorization of reactive orange 16 dye using a fabricated charcoal base metallic composite electrode, Malaysian J. Anal. Sci., 2015, 19, 493–502.
- N. Farfour, M. El Mahi Chbihi, D. Takky, K. Eddahaoui, S. Benmokhtar, Catalytic oxidation of methanol on Pt/X (X = CaTP, NaTP) electrodes in sulfuric acid solution, Mediterr. J. Chem., 2013, 2, 595–606.
- K. M. Hassan, A. A. Hathoot, R. Maher, M. Abdel Azzem, Electrocatalytic oxidation of ethanol at Pd, Pt, Pd/Pt and Pt/Pd nanoparticles supported on poly 1,8-diaminonaphthalene film in alkaline medium, RSC Adv., 2018, 8, 15417–15426.
- P. Zuman, Substituent Effects in Organic Polarography, Plenum Press, New York, NY, 1967.
- F. G. Thomas, K. G. Boto, The Chemistry of the Hydrazo, Azo and Azoxy Groups, John Wiley, London, 1975.
- D. K. Gosser, Cyclic voltammetry : simulation and analysis of reaction mechanisms, VCH, New York, 1993.
- M. S. Mahmoud, J. Y. Farah, T. E. Farrag, Enhanced removal of Methylene Blue by electrocoagulation using iron electrodes, Egypt. J. Pet., 2013, 22, 211–216.
DOI: http://dx.doi.org/10.13171/mjc10102001311108sea
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
