Increasing Stability of a-amylase Obtained from Bacillus subtilis ITBCCB148 by Immobilization with Chitosan
Abstract
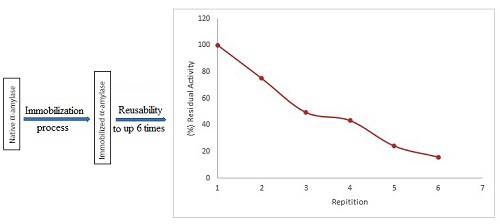
In this research, the immobilization of α-amylase from Bacillus subtilis ITBCCB148 by crosslinking method on chitosan matrix has been performed. This research aims to know the effect of immobilization on the thermal stability of α-amylase. The results showed that the native α-amylase has an optimum temperature of 65oC, KM = 1.6 mg mL-1 substrate, and Vmax = 39.7 µmol mL-1 min-1. The immobilized α-amylase has optimum temperature of 75oC, KM = 3.5 mg mL-1 substrate, and Vmax = 7.05 µmol mL-1 min-1. The residual activity of the native and immobilized enzyme on thermal stability test at 65oC for 80 minutes was 58% and 86.15%, respectively. The immobilized enzyme can be reused up to six repeated cycles.The thermodynamic data of native enzyme was t½ = 113.6 min, ki = 6.1x10-3 min-1, and ΔGi = 107.3 kJ mol-1, while the immobilized enzyme was t½ = 433.1 min, ki= 1.6x10-3 min-1, and ΔGi 111.1 kJ mol-1. Based on the decrease of ki, and the increase of ΔGi and half-life(t½) values, the immobilization of α-amylase with chitosan can increase the thermal stability of this enzyme.
Full Text:
PDFReferences
- V. Horvathova, S. Janecek, E. Sturdik, Amylolytic enzymes: Their specificities, origins, and properties, Biologia Bratislava, 2000, 55, 605-615.
- W. M. Fogarty, C. T. Kelly, Enzyme and Fermentation Biotechnology, Ellis Horwood Limited, West Sussex, England, 1979, pp, 45-52.
- A. A. Simair, A. S. Qureshi, I. Khushk, C. H. Ali, S. Lashari, M. A. Bhutto, C. Lu, Production and partial characterization of α-amylase enzyme from bacillus sp. bcc 01-50 and potential applications, BioMed Research International, 2017, 2017, 1-9.
- S. Trabelsi, S. B. Mabrouk, M. Kriaa, R. Ameri, M. Sahnoun, M. Mezghani, S. Bejar, The optimized production, purification, characterization, and application in the bread-making industry of three acid-stable alpha-amylases isoforms from a new isolated Bacillus subtilis strain US586, Journal of Food Biochemistry, 2019, 43(5), e12826.
- A. C. Yu, J. F. Loo, S. Yu, S. K. Kong, T. F. Chan, Monitoring bacterial growth using tunable resistive pulse sensing with a pore-based technique, Applied Microbiology and Biotechnology, 2014, 98, 855-862.
- R. Singh, M. Kumar, A. Mittal, P. K. Mehta, Microbial enzymes: industrial progress in 21st century, Biotech, 2016, 6, 174.
- G. Bayramoglu, M. Yilmaz, M. Y. Arica, Immobilization of a thermostable a-amylase onto reactive membranes: kinetics characterization and application to continuous starch hydrolysis, Food Chemistry, 2004, 84, 591-599.
- Yandri, T. Suhartati, S. Hadi, Immobilization of -amylase from locale bacteria isolate Bacillus subtilis ITBCCB148 with diethylaminoethyl cellulose (DEAE-Cellulose), Material Science Research India, 2010, 7, 123-128.
- Yandri, D. Susanti, T. Suhartati, S. Hadi, Immobilization of α-amylase from Locale Bacteria Isolate Bacillus subtilis ITBCCB148 with Carboxymethyl Cellulose (CM-Cellulose), Modern Applied Science, 2012, 6(3), 81-86.
- Yandri, T. Suhartati, S. D. Yuwono, H. I. Qudus, E. R. Tiarsa, S. Hadi, Immobilization of -amylase From Bacillus subtilis ITBCCB148 Using Bentonit, Asian Journal of Microbiology, Biotechnology and Environmental Science, 2018, 20, 487-492.
- M. E. Sedghat, M. Ghiaci, H. Aghaei, S. Soleimanian-Zad. Enzyme immobilization. Part 3 Immobilization of α-amylase on Na-bentonite and modified bentonite, Applied Clay Science, 2009, 46, 125-130.
- Yandri, P. Amalia, T. Suhartati, S. Hadi, Effect of immobilization towards thermal stability of α-amylase isolated from locale bacteria isolate Bacillus subtilis ITBCCB148 with calcium alginate, Asian Journal of Chemistry, 2013, 25, 6897-6899.
- P. Tripathi, A. Kumari, P. Rath, A. M. Kayastha, Immobilization of α-amylase from mung beans (Vigna radiata) on Amberlite MB 150 and chitosan beads: A comparative study, Journal of Molecular Catalysis B: Enzymatic, 2007, 49, 69–74.
- N. Jaiswal, O. Prakash, M. Talat, S. H. Hasan, α-Amylase immobilization on gelatin: Optimization of process variables, Journal of Genetic Engineering and Biotechnology, 2012, 10, 161-167.
- S. A. Cetinus, H. N. Oztop, Immobilization of catalase into chemically crosslinked chitosan beads, Enzyme and Microbial Technology, 2003, 32, 889-894.
- Yandri, T. Suhartati, S. Hadi, Purification and Characterization of Extracellular a-Amilase Enzyme from Locale Bacteria Isolate Bacillus subtilis ITBCCB148, European Journal of Scientific Research, 2010, 39, 64-67.
- B. K. Gogoi, R. L. Bezbaruah, K. R. Pillai, J. N. Baruah, Production, purification and characterization of an -amylase produced by Saccharomycopsis fibuligera, Journal of Applied Biochemistry, 1987, 63, 373-379.
- H. Fuwa, A new method for microdetermination of amylase activity by the use of amylose as the substrat, The Journal of Biochemistry, 1954, 41, 583-603.
- M. Mandels, R. Andreotti, C. Roche, Measurement of saccharifying cellulase, Biotechnology Bioengineering, 1976, 6, 21-33.
- O. H. Lowry, N. J. Rosebrough, A. L. Farr, R. J. Randall, Protein measurement with the Folin phenol reagent, Journal of Biological Chemistry, 1951, 193-265.
- B. Krajewska, Application of chitin- and chitosan-based materials for enzyme immobilization: a review, Enzyme and Microbial Technology, 2004, 35, 126-139.
- Z. Yang, D. Michael, A. Robert, X. Y. Fang, J. R. Alan, Polyethylene glycol-induced stabilization of subtilisin, Enzyme and Microbial Technology, 1996, 18, 82-89.
- D. Kazan, H. Ertan, A. Erarslan, Stabilization of Escherichia coli Penicillin G acylase against thermal inactivation by cross-linking with dextran dialdehyde polymers, Applied Microbiology and Biotechnology, 1997, 48, 191-197.
- M. Y. Chang, R. S. Juang, Activities, stabilities and reaction kinetics of three free and chitosan-clay composite immobilized enzymes, Enzyme and Microbial Technology, 2004, 36, 75-82.
- S. Stahl, Thermophilic microorganisms: The biological background for thermophily and thermoresistance of enzymes in Thermostability of Enzymes (Gupta, M.N. editor), Springer Verlag, New Delhi, 1999, pp. 59-60.
- M. T. Xavier, V. F. Soares, D. G. Freire, C. P. Moreira, M. F. Mendes, E. Bon, -Amylase and glucoamylase immobilized on chitin and ceramic supports, Biomass, 1987, 13, 25-32.
DOI: http://dx.doi.org/10.13171/mjc10202002131126ysh
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
