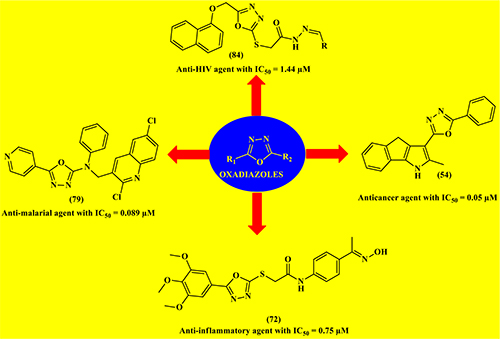
Recent advances on oxadiazole motifs: Synthesis, reactions and biological activities
Abstract
1,3,4-oxadiazole derivatives have shown to have diverse and vast applications, from medicinal chemistry for the treatment and possible treatment of various ailments to its application in the industrial development when used as corrosion inhibitions and light emitting diodes. These diverse applications can be as a result of the numerous viable synthetic pathways illustrated in this review. 1,3,4-oxadiazoles can be synthesized in very high yields, using green approaches and having various life-changing applications. This review explores the various recent synthetic routes available for the development of 1,3,4-oxadiazoles and their biological activities.
Full Text:
PDFReferences
- V. D. Aderohunmu, T. T. Odutola, O. O. Ajani, E. F. Adebiyi, Tosmic reagent: an excellent precursor in the synthesis of biologically active heterocycles, Rasayan J. Chem., 2019, 12, 1919-1926.
- O. O. Ajani, J. T. Isaac, T. F. Owoeye, A. A. Akinsiku, Exploration of the chemistry and biological properties of pyrimidine as a privilege pharmacophore in therapeutics, Int. J. Biol. Chem., 2015, 9, 148–177.
- X. F. Shang, Z. M. Zhao, J. C. Li, G. Z. Yang, Y. Q. Liu, L. X. Dai, Insecticidal and antifungal activities of Rheum palmatum L. anthraquinones and structurally related compounds, Ind. Crops Prod., 2019, 137, 508–520.
- S. A. Mayank, N. Kaur, N. Garg, N. Singh, Anticancer SAR establishment and novel accruing signal transduction model of drug action using biscoumarin scaffold, Comput. Biol. Chem., 2019, 83, 107104.
- W. T. Wang, H. Qian, J. W. Wu, X. W. Chen, J. Q. Li, Synthesis and antidepressant-like activity of novel alkoxy-piperidine derivatives targeting SSRI/5-HT1A/5-HT7, Bioorg. Med. Chem. Lett., 2019, 29, 126769.
- L. Huang, J. Ding, M. Li, Z. Hou, Y. Geng, X. Li, Discovery of [1,2,4]-triazolo [1,5-a]pyrimidine-7(4H)-one derivatives as positive modulators of GABAA1 receptor with potent anticonvulsant activity and low toxicity, Eur. J. Med. Chem., 2019, 111824.
http://dx.doi.org/10.1016/J.EJMECH.2019.111824.
- X. M. Chu, C. Wang, W. L. Wang, L. L Liang, W. Liu, K. K. Gong, Triazole derivatives and their antiplasmodial and antimalarial activities, Eur. J. Med. Chem., 2019, 166, 206–223.
- P. Pitasse-Santos, V. Sueth-Santiago, M. E. F. Lima, 1,2,4- and 1,3,4-Oxadiazoles As Scaffolds in the Development of Antiparasitic Agents, J. Braz. Chem. Soc., 2018, 29, 435–456.
- M. Carbone, Y. Li, C. Irace, E. Mollo, F. Castelluccio, A. Di Pascale, Structure and cytotoxicity of phidianidines A and B: First finding of 1,2,4-oxadiazole system in a marine natural product, Org. Lett., 2011, 13, 2516–2519.
- J. T. Brogan, S. L. Stoops, C. W. Lindsley, Total synthesis and biological evaluation of phidianidines a and b uncovers unique pharmacological profiles at CNS targets, ACS Chem. Neurosci., 2012, 3, 658–664.
- W. Caneschi, K. B. Enes, C. Carvalho de Mendonça, F. de Souza Fernandes, F. B. Miguel, J. M. da Silva, Synthesis and anticancer evaluation of new lipophilic 1,2,4 and 1,3,4-oxadiazoles, Eur. J. Med. Chem., 2019, 165, 18–30.
- X. Song, P. Li, M. Li, A. Yang, L. Yu, L. Luo, Synthesis and investigation of the antibacterial activity and action mechanism of 1,3,4-oxadiazole thioether derivatives, Pestic. Biochem. Physiol., 2018, 147, 11–19.
- A. Zumla, A. George, V. Sharma, R. H. N. Herbert, A. Oxley, M. Oliver, The WHO 2014 Global tuberculosis report—further to go, Lancet Glob. Heal., 2015, 3, e10–e12.
- S. A. Popov, M. D. Semenova, D. S. Baev, I. V. Sorokina, N. A. Zhukova, T. S. Frolova, Lupane-type conjugates with aminoacids, 1,3,4- oxadiazole and 1,2,5-oxadiazole-2-oxide derivatives: Synthesis, anti-inflammatory activity and in silico evaluation of target affinity, Steroids, 2019, 150, 108443.
- X. Gan, D. Hu, Z. Chen, Y. Wang, B. Song, Synthesis and antiviral evaluation of novel 1,3,4-oxadiazole/thiadiazole-chalcone conjugates, Bioorg. Med. Chem. Lett, 2017, 27, 4298–4301.
- Y. Zhang, C. Zuniga, S. J. Kim, D. Cai, S. Barlow, S. Salman, Polymers with carbazole-oxadiazole side chains as ambipolar hosts for phosphorescent light-emitting diodes, Chem. Mater., 2011, 23, 4002–4015.
- A. Bruno, C. Borriello, T. Di Luccio, L. Sessa, S. Concilio, S. A. Haque, Oxadiazole-carbazole polymer (POC)-Ir(ppy)3 tunable emitting composites, Opt. Mater. (Amst), 2017, 66, 166–170.
- D. Zhang, X. Hua, M. Liu, C. Wu, W. Wei, Y. Liu, Design, synthesis and herbicidal activity of novel sulfonylureas containing triazole and oxadiazole moieties, Chem. Res. Chinese Univ., 2016, 32, 607–614.
- P. A. Rugmini, M. Prajila, A. Joseph, Effect of substitution and temperature on the corrosion inhibition properties of benzimidazole bearing 1, 3, 4-oxadiazoles for mild steel in sulphuric acid: Physicochemical and theoretical studies, J. Environ. Chem. Eng., 2018, 6, 1072–1085.
- M. Bouanis, M. Tourabi, A. Nyassi, A. Zarrouk, C. Jama, F. Bentiss, Corrosion inhibition performance of 2,5-bis(4-dimethylaminophenyl)-1,3,4-oxadiazole for carbon steel in HCl solution: Gravimetric, electrochemical and XPS studies, Appl. Surf. Sci., 2016, 389, 952–966.
- R. Sharma, N. Kumar, R. Yaday, Chemistry and Pharmacological Importance of 1,3,4-oxadiazole Derivatives, Res. Rev. J. Chem., 2015, 4, 1–27.
- Nagaraj, K. C. Chaluvaraju, M. S. Niranjan, S. Kiran. 1, 3, 4 Oxadiazole a Potent Drug Candidate With Various Pharmacological Activities, Int. J. Pharm. Pharm. Sci., 2011, 3, 9–16.
- S. Sharma, P. K. Sharma, N. Kumar, R. Dudhe, Chemistry and pharmacological potentials of Oxadiazoles: A Review, Der Pharma Chem., 2010, 2, 253–263.
- A. D. M. Curtis, N. Jennings, 1,2,4-Triazoles. Compr. Heterocycl, Chem. 2008, 3, 159–209. http://dx.doi.org/10.1016/B978-008044992-0.00502-2.
- A. Almasirad, S. A. Tabatabai, M. Faizi, A. Kebriaeezadeh, N. Mehrabi, A. Dalvandi, Synthesis and anticonvulsant activity of new 2-substituted-5- [2-(2-fluorophenoxy)phenyl]-1,3,4-oxadiazoles and 1,2,4-triazoles, Bioorg. Med. Chem. Lett., 2004, 14, 6057–6059.
- A. Kakefuda, T. Suzuki, T. Tobe, A. Tahara, S. Sakamoto, S. Tsukamoto, Discovery of 4,5-Diphenyl-1,2,4-triazole Derivatives as a Novel Class of Selective Antagonists for the Human V1A Receptor, Bioorg. Med. Chem., 2002, 10, 1905–1912.
- S. A. Khanum, S. Shashikanth, S. Umesha, R. Kavitha, Synthesis and antimicrobial study of novel heterocyclic compounds from hydroxybenzophenones, Eur. J. Med. Chem., 2005, 40, 1156–1162.
- Y. Nagao, T. Hirata, S. Goto, S. Sano, A. Kakehi, K. Lizuka, Intramolecular nonbonded S···O interaction recognized in (acylimino)thiadiazoline derivatives as angiotensin II receptor antagonists and related compounds, J. Am. Chem. Soc., 1998, 120, 3104–3110.
- M. R. Grimmett, I. Brian, Synthesis and Reactions of lithiated monocyclic azoles containing two or more hetero-atoms. Part vi: Triazoles, tetrazoles, oxadiazoles, and thiadiazoles, Heterocycles, 1995, 41, 1525–1574. http://dx.doi.org/10.3987/REV-95-468.
- K. Ohmoto, T. Yamamoto, M. Okuma, T. Horiuchi, H. Imanishi, Y. Odagaki, Development of orally active nonpeptidic inhibitors of human neutrophil elastase, J. Med. Chem., 2001, 44, 1268–1285.
- A. A. Tolmachev, E. V. Zarudnitskii, A. A. Yurchenko, A. M. Pinchuk, Phosphorylation of 2-phenyl-1,3,4-oxadiazole with phosphorus (III) halides, Chem. Heterocycl. Compd., 1999, 35, 1117–1119.
- S. G. Narella, M. G. Shaik, A. Mohammed, M. Alvala, A. Angeli, C. T. Supuran, Synthesis and biological evaluation of coumarin-1,3,4-oxadiazole hybrids as selective carbonic anhydrase IX and XII inhibitors, Bioorg. Chem., 2019, 87, 765–772.
- P. Mishra, P. Sharma, P. N. Tripathi, S. K. Gupta, P. Srivastava, A. Seth, Design and development of 1,3,4-oxadiazole derivatives as potential inhibitors of acetylcholinesterase to ameliorate scopolamine-induced cognitive dysfunctions, Bioorg. Chem., 2019, 89, 103025.
- K. Lakshmithendral, K. Saravanan, R. Elancheran, K. Archana, N. Manikandan, H. A. Arjun, Design, synthesis and biological evaluation of 2-(phenoxymethyl)-5-phenyl-1,3,4-oxadiazole derivatives as anti-breast cancer agents, Eur. J. Med. Chem., 2019, 168, 1–10.
- S. Bala, S. Kamboj, A. Kajal, V. Saini, D. N. Prasad, Antimicrobial Potential , and Computational Studies, Biomed Res, Int., 2014.
- Q. Gao, S. Liu, X. Wu, J. Zhang, A. Wu, Direct Annulation of Hydrazides to 1,3,4-Oxadiazoles via Oxidative C(CO)-C(Methyl) Bond Cleavage of Methyl Ketones, Org. Lett., 2015, 17, 2960–2963.
- Y. Fan, Y. He, X. Liu, T. Hu, H. Ma, X. Yang, Iodine-Mediated Domino Oxidative Cyclization: One-Pot Synthesis of 1,3,4-Oxadiazoles via Oxidative Cleavage of C(sp2)–H or C(sp)–H Bond, J. Org. Chem., 2016, 81, 6820–6825.
- A. Kudelko, K. Jasiak, K. Ejsmont, Study on the synthesis of novel 5-substituted 2-[2-(pyridyl)ethenyl]-1,3,4-oxadiazoles and their acid-base interactions, Monatshefte fur Chemie., 2015, 146, 303–311.
- G. Prabhu, V. V. Sureshbabu, Hypervalent iodine(V) mediated mild and convenient synthesis of substituted 2-amino-1,3,4-oxadiazoles, Tetrahedron Lett., 2012, 53, 4232–4234.
- T. Fang, Q. Tan, Z. Ding, B. Liu, B. Xu, Pd-Catalyzed Oxidative Annulation of Hydrazides with Isocyanides: Synthesis of 2-Amino-1,3,4-oxadiazoles, Org. Lett., 2014, 16, 2342–2345.
- S. N. R. Mule, S. K. Battula, G. Velupula, D. R. Guda, H. B. Bollikolla, 10-Camphorsulfonic acid ((±)-CSA) catalyzed facile one-pot synthesis of a new class of 2,5-disubstituted 1,3,4-oxadiazoles, RSC Adv., 2014, 4, 58397–58403.
- F. Yu, A. Guan, M. Li, L. Hu, X. Li, Design, synthesis, and fungicidal activity of novel 1,3,4-oxadiazole derivatives, Chinese Chem. Lett., 2018, 29, 915–918.
- B. Koçyiğit-Kaymakçıoğlu, E. E. Oruç-Emre, S. Ünsalan, N. Tabanca, S. I. Khan, D. E. Wedge, Synthesis and biological activity of hydrazide-hydrazones and their corresponding 3-Acetyl-2,5-disubstituted-2,3-dihydro-1,3,4-oxadiazoles, Med. Chem. Res., 2012, 21, 3499–3508.
- L. Zhang, X. Zhao, X. Jing, X. Zhang, S. Lü, L. Luo, Oxidation of sp3 CH bonds in N-alkylhydrazides: Access to 2,5-disubstituted 1,3,4-oxadiazole derivatives, Tetrahedron Lett., 2016, 57, 5669–5672.
- G. Zhang, Y. Yu, Y. Zhao, X. Xie, C. Ding, Iron(III)/TEMPO-Catalyzed Synthesis of 2,5-Disubstituted 1,3,4-Oxadiazoles by Oxidative Cyclization under Mild Conditions, Synlett, 2017, 28, 1373–1377.
- G. Majji, S. K. Rout, S. Guin, A. Gogoi, B. K. Patel, Iodine-catalysed oxidative cyclisation of acylhydrazones to 2,5-substituted 1,3,4-oxadiazoles, RSC Adv., 2014, 4, 5357–5362.
- S. J. Yang, S. H. Lee, H. J. Kwak, Y. D. Gong. Regioselective Synthesis of 2-Amino-Substituted 1,3,4-Oxadiazole and 1,3,4-Thiadiazole Derivatives via Reagent-Based Cyclization of Thiosemicarbazide Intermediate, J. Org. Chem, 2013, 78, 438–444.
- S. Maghari, S. Ramezanpour, F. Darvish, S. Balalaie, F. Rominger, H. R. Bijanzandeh, A new and efficient synthesis of 1,3,4-oxadiazole derivatives using TBTU, Tetrahedron, 2013, 69, 2075–2080.
- S. J. Yang, J. H. Choe, A. Abdildinova, Y. D. Gong, A Highly Efficient Diversification of 2-Amino/Amido-1,3,4-oxadiazole and 1,3,4-Thiadiazole Derivatives via Reagent-Based Cyclization of Thiosemicarbazide Intermediate on Solid-Phase ACS Comb. Sci., 2015, 17, 732–741.
- A. Abdildinova, S. J. Yang, Y. D. Gong. Solid-phase parallel synthesis of 1,3,4-oxadiazole based peptidomimetic library as a potential modulator of protein-protein interactions, Tetrahedron, 2018, 74, 684–691.
- M. A. Rahman, M. R. Karim, M. Arifuzzaman, T. A. Siddiquee, A. H. Mirza, ZnCl2 catalyzed efficient synthesis of 1,3,4-oxadiazole and 1,3,4-thiadiazole, Tetrahedron Lett., 2014, 55, 3267–3273.
- Y. Xie, J. Liu, P. Yang, X. Shi, J. Li, Synthesis of 2-amino-1,3,4-oxadiazoles from isoselenocyanates via cyclodeselenization, Tetrahedron, 2011, 67, 5369–5374.
- L. Santhosh, C. Srinivasulu, S. Durgamma, G. Prabhu, V. V. Sureshbabu, Facile one-pot synthesis of 2-amino-1,3,4-oxadiazole tethered peptidomimetics by molecular-iodine-mediated cyclodeselenization, New J. Chem., 2017, 41, 11225–11229.
- M. Rouhani, A. Ramazani, S. W. Joo, Novel, fast and efficient one-pot sonochemical synthesis of 2-aryl-1,3,4-oxadiazoles, Ultrason. Sonochem., 2014, 21, 262–267.
- M. Rouhani, A. Ramazani, S. W. Joo, Ultrasonics in isocyanide-based multicomponent reactions: A new, efficient and fast method for the synthesis of fully substituted 1,3,4-oxadiazole derivatives under ultrasound irradiation, Ultrason. Sonochem., 2015, 22, 391–396.
- A. K. Yadav, L. D. S. Yadav, Visible-light-promoted aerobic oxidative cyclization to access 1,3,4-oxadiazoles from aldehydes and acylhydrazides, Tetrahedron Lett., 2014, 55, 2065–2069.
- P. Diao, Y. Ge, W. Zhang, C. Xu, N. Zhang, C. Guo, Synthesis of 2,5-disubstituted 1,3,4-oxadiazoles by visible-light-mediated decarboxylation–cyclization of hydrazides and diketones, Tetrahedron Lett., 2018, 59, 767–770.
- L. Wang, Y. Wang, Q. Chen, M. He, Photocatalyzed facile synthesis of 2,5-diaryl 1,3,4-oxadiazoles with polyaniline- g-C3N4-TiO2 composite under visible light, Tetrahedron Lett., 2018, 59, 1489–1492.
- P. Stabile, A. Lamonica, A. Ribecai, D. Castoldi, G. Guercio, O. Curcuruto, Mild and convenient one-pot synthesis of 1,3,4-oxadiazoles, Tetrahedron Lett., 2010, 51, 4801–4805.
- N. Mihailović, V. Marković, I. Z. Matić, N. S. Stanisavljević, Ž. S. Jovanović, S. Trifunović, Synthesis and antioxidant activity of 1,3,4-oxadiazoles and their diacylhydrazine precursors derived from phenolic acids, RSC Adv., 2017, 7, 8550–8560.
- S. Wet-osot, W. Phakhodee, M. Pattarawarapan, Application of N-Acylbenzotriazoles in the Synthesis of 5-Substituted 2-Ethoxy-1,3,4-oxadiazoles as Building Blocks toward 3,5-Disubstituted 1,3,4-Oxadiazol-2(3H)-ones, J. Org. Chem., 2017, 82, 9923–9929.
- C. X. Guo, W. Z. Zhang, N. Zhang, X. B. Lu, 1,3-Dipolar Cycloaddition of Nitrile Imine with Carbon Dioxide: Access to 1,3,4-Oxadiazole-2(3H)-ones, J. Org. Chem., 2017, 82, 7637–7642.
- L. Wang, J. Cao, Q. Chen, M. He, One-Pot Synthesis of 2,5-Diaryl 1,3,4-Oxadiazoles via Di-tert-butyl Peroxide Promoted N-Acylation of Aryl Tetrazoles with Aldehydes, J. Org. Chem., 2015, 80, 4743–4748.
- N. C. Desai, H. Somani, A. Trivedi, K. Bhatt, L. Nawale, V. M. Khedkar, Synthesis, biological evaluation and molecular docking study of some novel indole and pyridine based 1,3,4-oxadiazole derivatives as potential antitubercular agents, Bioorg. Med. Chem. Lett., 2016, 26, 1776–1783.
- S. Singh, L. K. Sharma, A. Saraswat, I. R. Siddiqui, H. K. Kehri, R. K. P. Singh, Electrosynthesis and screening of novel 1,3,4-oxadiazoles as potent and selective antifungal agents, RSC Adv., 2013, 3, 4237–4245.
- S. Bajaj, P. P. Roy, J. Singh, Synthesis, thymidine phosphorylase inhibitory and computational study of novel 1,3,4-oxadiazole-2-thione derivatives as potential anticancer agents, Comput. Biol. Chem., 2018, 76, 151–160.
- J. D. S. Chaves, L. G. Tunes, C. H. Franco, T. M. Francisco, C. C. Corrêa, S. M. F. Murta, Novel gold(I) complexes with 5-phenyl-1,3,4-oxadiazole-2-thione and phosphine as potential anticancer and antileishmanial agents, Eur. J. Med. Chem., 2017, 127, 727–739.
- V. Nieddu, G. Pinna, I. Marchesi, L. Sanna, B. Asproni, G. A. Pinna, Synthesis and Antineoplastic Evaluation of Novel Unsymmetrical 1,3,4-Oxadiazoles, J. Med. Chem., 2016, 59, 10451–10469.
- B. Mochona, X. Qi, S. Euynni, D. Sikazwi, N. Mateeva, K. F. Soliman, Design and evaluation of novel oxadiazole derivatives as potential
prostate cancer agents, Bioorg. Med. Chem. Lett., 2016, 26, 2847–2851.
- E. M. M. Gamal, M. I. El-Gamal, M. S. Abdel-Maksoud, K. H. Yoo, C. H. Oh, Synthesis and in vitro antiproliferative activity of new 1,3,4-oxadiazole derivatives possessing sulfonamide moiety, Eur. J. Med. Chem., 2015, 90, 45–52.
- A. V. R. Subba, V. Vishnu, R. N. V. Subba, R. T. Srinivasa, S. P. Shaik, C. Bagul, Synthesis and biological evaluation of imidazopyridinyl-1,3,4-oxadiazole conjugates as apoptosis inducers and topoisomerase IIα inhibitors, Bioorg. Chem., 2016, 69, 7–19.
- N. J. P. Subhashini, B. Bhadraiah, P. Janaki, Synthesis and biological evaluation of 1,3,4-oxadiazole fused pyridine derivatives as antibacterial and antifungal agents, Russ. J. Gen. Chem., 2017, 87, 550–553.
- G. A. Ç. Karaburun, G. B. K. Çavuşo, U. A. Çevik, D. Osmaniye, G. B. N. Sa, S. Levant, Synthesis and antifungal potential of some novel benzimidazole-1,3,4-oxadiazole compounds, Molecules, 2019, 24, 1–14.
- X. Wang, H. Hu, X. Zhao, M. Chen, T. Zhang, C. Geng, Novel quinazolin-4(3H)-one derivatives containing a 1,3,4-oxadiazole thioether moiety as potential bactericides and fungicides: Design, synthesis, characterization and 3D-QSAR analysis, J. Saudi Chem. Soc., 2019. doi:10.1016/J.JSCS.2019.07.006.
- J. Dai, W. Dan, S. Ren, C. Shang, J. Wang, Design, synthesis and biological evaluations of quaternization harman analogues as potential antibacterial agents, Eur. J. Med. Chem., 2018, 160, 23–36.
- X. Hua, Q. Yang, W. Zhang, Z. Dong, S. Yu, S. Schwarz, Antibacterial Activity and Mechanism of Action of Aspidinol Against Multi-Drug-Resistant Methicillin-Resistant Staphylococcus aureus, Front. Pharmacol., 2018, 9.
- Y. Guo, T. Xu, C. Bao, Z. Liu, J. Fan, R. Yang, Design and synthesis of new norfloxacin-1,3,4-oxadiazole hybrids as antibacterial agents against methicillin-resistant Staphylococcus aureus (MRSA), Eur. J. Pharm. Sci., 2019, 136, 104966.
- N. N. Farshori, A. Rauf, M. A. Siddiqui, E. S. Al-Sheddi, M. M. Al-Oqail, A facile one-pot synthesis of novel 2,5-disubstituted-1,3,4-oxadiazoles under conventional and microwave conditions and evaluation of their in vitro antimicrobial activities, Arab. J. Chem., 2017, 10, S2853–S2861.
- Beena, D. S. Rawat, Antituberculosis Drug Research: A Critical Overview. Med. Res. Rev., 2013, 33, 693–764.
- T. Kaneko, C. Cooper, K. Mdluli, Challenges and opportunities in developing novel drugs for TB, Future Med. Chem., 2011, 3, 1373–1400.
- U. Sharma, Current possibilities and unresolved issues of drug target validation in Mycobacterium tuberculosis, Expert Opin. Drug Discov., 2011, 6, 1171–1186.
- S. T. Dhumal, A. R. Deshmukh, M. R. Bhosle, V. M. Khedkar, L. U. Nawale, D. Sarkar, Synthesis and antitubercular activity of new 1,3,4-oxadiazoles bearing pyridyl and thiazolyl scaffolds, Bioorg. Med. Chem. Lett., 2016, 26, 3646–3651.
- A. N. Ambhore, S. S. Kamble, S. N. Kadam, R. D. Kamble, M. J. Hebade, S. V. Hese, Design, synthesis and in silico study of pyridine based 1,3,4-oxadiazole embedded hydrazinecarbothioamide derivatives as potent anti-tubercular agent, Comput. Biol. Chem., 2019, 80, 54–65.
- G. Karabanovich, J. Němeček, L. Valášková, A. Carazo, K. Konečná, J. Stolaříková, S-substituted 3,5-dinitrophenyl 1,3,4-oxadiazole-2-thiols and tetrazole-5-thiols as highly efficient antitubercular agents, Eur. J. Med. Chem., 2017, 126, 369–383.
- H. S. Abd-Ellah, M. Abdel-Aziz, M. E. Shoman, E. A. M Beshr, T. Kaoud, A. S. F. F. Ahmed, New 1,3,4-oxadiazole/oxime hybrids: Design, synthesis, anti-inflammatory, COX inhibitory activities and ulcerogenic liability, Bioorg. Chem., 2017, 74, 15–29.
- H. S. Abd-Ellah, M. Abdel-Aziz, M. E. Shoman, E. A. M, Beshr, T. S. Kaoud, A. S. F. F. Ahmed, Novel 1,3,4-oxadiazole/oxime hybrids: Synthesis, docking studies and investigation of anti-inflammatory, ulcerogenic liability and analgesic activities, Bioorg. Chem., 2016, 69, 48–63.
- A. G. Banerjee, N. Das, S. A. Shengule, R. S. Srivastava, S. K. Shrivastava, Synthesis, characterization, evaluation and molecular dynamics studies of 5, 6–diphenyl–1,2,4–triazin–3(2H)–one derivatives bearing 5–substituted 1,3,4–oxadiazole as potential anti-inflammatory and analgesic agents, Eur. J. Med. Chem., 2015, 101, 81–95.
- C. R. Biju, K. Ilango, M. Prathap, K. Rekha, Design and Microwave-assisted Synthesis of 1,3,4-Oxadiazole Derivatives for Analgesia and Anti-inflammatory Activity, J. Young Pharm., 2012, 4, 33–37.
- S. S. Thakkar, P. Thakor, H. Doshi, A. Ray, 1,2,4-Triazole and 1,3,4-oxadiazole analogues: Synthesis, MO studies, in silico molecular docking studies, antimalarial as DHFR inhibitor and antimicrobial activities, Bioorg. Med. Chem., 2017, 25, 4064–4075.
- G. Verma, M. F. Khan, L. N. Mohan, M. Ishaq, M. Akhter, A. Bakht, Targeting malaria and leishmaniasis: Synthesis and pharmacological evaluation of novel pyrazole-1,3,4-oxadiazole hybrids, Part II, Bioorg. Chem., 2019, 89, 102986.
- G. G. Ladani, M. P. Patel, Novel 1,3,4-oxadiazole motifs bearing a quinoline nucleus: synthesis, characterization and biological evaluation of their antimicrobial, antitubercular, antimalarial and cytotoxic activities, New J. Chem., 2015, 39, 9848–9857.
- S. K. Avula, A. Khan, N. U. Rehman, M. U. Anwar, Z. Al-Abri, A. Wadood, Synthesis of 1H-1,2,3-triazole derivatives as new α-glucosidase inhibitors and their molecular docking studies, Bioorg. Chem., 2018, 81, 98–106.
- M. J. Naim, M. J. Alam, F. Nawaz, V. G. M. Naidu, S. Aaghaz, M. Sahu, Synthesis, molecular docking and anti-diabetic evaluation of 2,4-thiazolidinedione based amide derivatives, Bioorg. Chem., 2017, 73, 24–36.
- M. J. Naim, O. Alam, M. J. Alam, M. Q. Hassan, N. Siddiqui, V. G. M. Naidu, Design, synthesis and molecular docking of thiazolidinedione based benzene sulphonamide derivatives containing pyrazole core as potential anti-diabetic agents, Bioorg. Chem., 2018, 76, 98–112.
- R. Bhutani, D. Pathak, G. Kapoor, A. Husain, M. A. Iqbal, Novel hybrids of benzothiazole-1,3,4-oxadiazole-4-thiazolidinone: Synthesis, in silico ADME study, molecular docking and in vivo anti-diabetic assessment, Bioorg. Chem., 2019, 83, 6–19.
- P. C. Shyma, B. Kalluraya, S. K. Peethambar, A. M. Vijesh, Synthesis, characterization, antidiabetic and antioxidant activity of 1,3,4-oxadiazole derivatives bearing 6-methyl pyridine moiety, Der Pharma Chem., 2015, 7, 137–145.
- S. Kumar, D. S. Rathore, G. Garg, K. Khatri, R. Saxena, S. K. Sahu, Synthesis and evaluation of some 2-((benzothiazol-2-ylthio) methyl)-5-phenyl-1, 3, 4-oxadiazole derivatives as antidiabetic agents, Asian Pacific J. Heal. Sci., 2017, 3, 65–74.
- W. A. El-Sayed, F. A. El-Essawy, O. M. Ali, B. S. Nasr, M. M. Abdalla, A. A. H. Abdel-Rahman, Anti-HIV Activity of New Substituted 1,3,4-Oxadiazole Derivatives and their Acyclic Nucleoside Analogues, Zeitschrift für Naturforsch., 2009, 64c, 773–778.
- W. Wu, Q. Chen, A. Tai, G. Jiang, G. Ouyang, Synthesis and antiviral activity of 2-substituted methylthio-5-(4-amino-2-methylpyrimidin-5-yl)-1,3,4-oxadiazole derivatives, Bioorg. Med. Chem. Lett., 2015, 25, 2243–2246.
-T. M. C. Tan, Y. Chen, K. H. Kong, J, Bai, Y. Li, S. G. Lim, Synthesis and the biological evaluation of 2-benzenesulfonylalkyl-5-substituted-sulfanyl-[1,3,4]-oxadiazoles as potential anti-hepatitis B virus agents, Antiviral Res., 2006, 71, 7–14.
-M. Ding, J. Fan, Q. Zhang, X. Wang, Y. Zhao, M. Huang, Semisynthesis and insecticidal activity of some novel fraxinellone-based thioethers containing 1,3,4-oxadiazole moiety, R. Soc. Open Sci., 2017, 4, 171053.
-Y. Guo, L. Qu, X. Wang, M. Huang, L. Jia, Y. Zhang, Iodine-catalyzed oxidative cyclisation for the synthesis of sarisan analogues containing 1,3,4-oxadiazole as insecticidal agents, RSC Adv., 2016, 6, 93505–93510.
-F. Z. Xu, Y .Y. Wang, D. X. Luo, G. Yu, S. X. Guo, H. Fu, Design, synthesis, insecticidal activity and 3D-QSR study for novel trifluoromethyl pyridine derivatives containing a 1,3,4-oxadiazole moiety, RSC Adv., 2018, 8, 6306–6314.
-Q. Liu, K. Chen, Q. Wang, J. Ni, Y. Li, H. Zhu, Synthesis, insecticidal activity, structure-activity relationship (SAR) and density functional theory (DFT) of novel anthranilic diamides analogs containing 1,3,4-oxadiazole rings, RSC Adv., 2014, 4, 55445–55451.
-Y. Li, H. Zhu, K. Chen, R. Liu, A. Khallaf, X. Zhang, Synthesis, insecticidal activity, and structure–activity relationship (SAR) of anthranilic diamides analogs containing oxadiazole rings, Org. Biomol. Chem., 2013, 11, 3979–3988.
-B. N. P. Kumar, K. N. Mohana, L. Mallesha, Synthesis of N-[{5-Aryl-1,3,4-oxadiazole-2-yl}methyl]-4-methoxyaniline Derivatives and Their Anticonvulsant Activity, Hindawi Publ. Corp. J. Chem., 2013, 2013, 1–8.
-S. A. Tabatabai, S. Lashkari, M. Zarrindast, M. Gholinbeikian, A. Shafiee, Design, Synthesis and Anticonvulsant Activity of 2- ( 2-Phenoxy ) phenyl-1,3,4-oxadiazole Derivatives, Iran. J. Pharm. Res., 2013, 12, 105–111.
-P. Singh, P. K. Sharma, J. K. Sharma, A. Upadhyay, N. Kumar, Synthesis and evaluation of substituted diphenyl-1,3,4-oxadiazole derivatives for central nervous system depressant activity, Org. Med. Chem. Lett., 2012, 2, 8.
-M. Pardo, E. Abrial, R. S. Jope, E. Beurel, GSK3β isoform-selective regulation of depression, memory and hippocampal cell proliferation, Genes, Brain Behav., 2016, 15, 348–355.
-E. J. Crofton, M. N. Nenov, Y. Zhang, F. Scala, S. A. Page, D. L. McCue, Glycogen synthase kinase 3 beta alters anxiety-, depression-, and addiction-related behaviors and neuronal activity in the nucleus accumbens shell, Neuropharmacology, 2017, 117, 49–60.
-M. A. Tantray, I. Khan, H. Hamid, M. S. Dhulap, A. Kalam, Synthesis of benzimidazole-based 1,3,4-oxadiazole-1,2,3-triazole conjugates as glycogen synthase kinase-3β inhibitors with antidepressant activity in vivo models, RSC Adv., 2016, 6, 43345–43355.
-M. A. Tantray, I. Khan, H. Hamid, M. S. Alam, A. Dhulap, A. Kalam, Synthesis of benzimidazole-linked-1,3,4-oxadiazole carboxamides as GSK-3β inhibitors with in vivo antidepressant activity. Bioorg. Chem., 2018, 77, 393–401.
-M. Kalhor, A. Dadras, Synthesis, Characterization, and Herbicidal Activities of New 1,3,4-Oxadiazoles, 1,3,4-Thiadiazoles, and 1,2,4-Triazoles Derivatives Bearing (R)-5-Chloro-3-fluoro-2-phenoxypyridine, J. Heterocycl. Chem., 2013, 50, 220–224.
-G. X. Sun, M. Y. Yang, Z. H. Sun, H. K. Wu, X. H Liu, Y. Y. Wei, Synthesis and bioactivities of novel 1,3,4-oxadiazole derivatives containing 1,2,3-thiadiazole moiety, Phosphorus. Sulfur. Silicon Relat. Elem., 2014, 189, 1895–1900.
-K. Dasuri, L. Zhang, J. N. Keller, Oxidative stress, neurodegeneration, and the balance of protein degradation and protein synthesis, Free Radic. Biol. Med., 2013, 62, 170–185.
-Y. Ünver, H. Gökce, E. Bektaş, F. Çelik, İ. Değirmencioğlu, New bis 1,3,4-oxadiazole derivatives: syntheses, characterizations, computational studies, and antioxidant activities, Can. J. Chem., 2018, 96, 1047–1059.
-A. C. Sauer, J. G. Leal, S. T. Stefanello, M. T. B. Leite, M. B. Souza, F. A. A. Soares, Synthesis and antioxidant properties of organosulfur and organoselenium compounds derived from 5-substituted-1,3,4-oxadiazole/thiadiazole-2-thiols, Tetrahedron Lett., 2017, 58, 87–91.
-L. Ma, Y. Xiao, C. Li, Z. L. Xie, D. D. Li, Y. T. Wang, Synthesis and antioxidant activity of novel Mannich base of 1,3,4-oxadiazole derivatives possessing 1,4-benzodioxan, Bioorg. Med. Chem., 2013, 21, 6763–6770.
-Y. Kotaiah, N. Harikrishna, K. Nagaraju, R. C. Venkata, Synthesis and antioxidant activity of 1,3,4-oxadiazole tagged thieno[2,3-d]pyrimidine derivatives, Eur. J. Med. Chem., 2012, 58, 340–345.
-A. Cavalli, M. L. Bolognesi, Neglected Tropical Diseases: Multi-Target-Directed Ligands in the Search for Novel Lead Candidates against Trypanosoma and Leishmania, J. Med. Chem., 2009, 52, 7339–7359.
-M. Taha, N. H. Ismail, S. Imran, E. H. Anouar, M. Selvaraj, W. Jamil, Synthesis and molecular modelling studies of phenyl linked oxadiazole-phenylhydrazone hybrids as potent antileishmanial agents, Eur. J. Med. Chem., 2017, 126, 1021–1033.
-M. Taha, N. H. Ismail, M. Ali, U. Rashid, S. Imran, N. Uddin, Molecular hybridization conceded exceptionally potent quinolinyl-oxadiazole hybrids through phenyl linked thiosemicarbazide antileishmanial scaffolds: In silico validation and SAR studies, Bioorg. Chem., 2017, 71, 192–200.
-J. Kaur, M. Soto-Velasquez, Z. Ding, A. Ghanbarpour, M. A. Lill, R. M. van Rijn, Optimization of a 1,3,4-oxadiazole series for inhibition of Ca2+/calmodulin-stimulated activity of adenylyl cyclases 1 and 8 for the treatment of chronic pain, Eur. J. Med. Chem., 2019, 162, 568–585.
-P. N. Tripathi, P. Srivastava, P. Sharma, A. Seth, S. K. Shrivastava, Design and development of novel N-(pyrimidin-2-yl)-1,3,4-oxadiazole hybrids to treat cognitive dysfunctions, Bioorg. Med. Chem., 2019, 27, 1327–1340.
-S. A. Shahzad, M. Yar, M. Bajda, L. Shahzadi, Z. A. Khan, S. A. R. Naqvi, Synthesis, thymidine phosphorylase inhibition and molecular modeling studies of 1,3,4-oxadiazole-2-thione derivatives, Bioorg. Chem., 2015, 60, 37–41.
DOI: http://dx.doi.org/10.13171/mjc10502005121200ooa
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
