A new percentage of biphasic calcium phosphate (BCP) of nanoparticles synthesized directly by hydrothermal process
Abstract
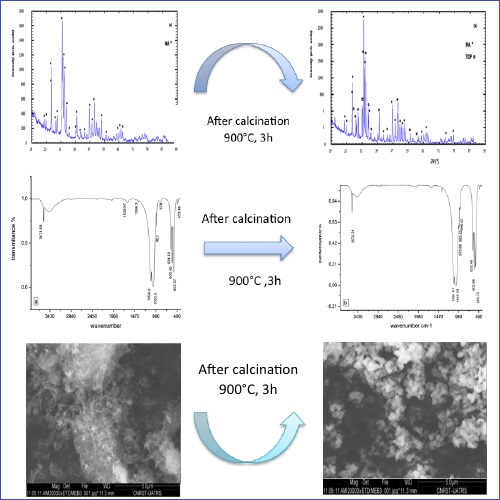
Apatitic calcium phosphates have a mineral phase close to that of bone and are commonly used as substitutes or fillers in bone surgery. Also, they are used for their excellent biocompatibility and bioactivity. The purpose of this study is to produce directly biphasic calcium phosphate (BCP) from calcium-deficient apatite (CDHA), by the hydrothermal process. The precursors used were calcium acetate (CH3COO)2 Ca, phosphoric acid (H3PO4) and triethylamine N (CH2CH3)3, the product was calcined at 900°C. The composition phase, morphology, particle size and the molecular structure of the product were studied using these techniques: X-Ray Diffraction techniques, Scanning Electron Microscopy (SEM), Transmission Electron Microscopy, Fourier Transforms Infrared spectroscopy (FT-IR ) to reveal its phase content, Inductively Coupled Plasma-Atomic Emission Spectrometers (ICP-AES), Thermogravimetric analysis, Thermo-Differentials (ATG / ATD). The results showed that the synthesized product is CDHA. It gives after calcination at 900°C, the biphasic calcium phosphate (a mixture of HA + β-TCP). The product is also found with a new percentage and high crystallinity and nanometric size.
Full Text:
PDFReferences
- J. A. Stammeier, B. Purgstaller, D. Hippler, V. Mavromatis, M. Dietzel, In-situ Raman spectroscopy of amorphous calcium phosphate to crystalline hydroxyapatite transformation, MethodsX, 2018, 5, 1241–1250.
- D. S. Metsger, T. D. Driskell, J. R. Paulsrud, Tricalcium phosphate ceramic—a resorbable bone implant: review and current status, J Am Dent Assoc, 1982, 105, 1035–1038.
- W. Habraken, P. Habibovic, M. Epple, M. Bohner, Calcium phosphates in biomedical applications: materials for the future?, Mater Today, 2016, 19, 69–87.
- D.-M. Liu, H.-M. Chou, Formation of a new bioactive glass-ceramic, J Mater Sci Mater Med, 1994, 5, 7–10.
- R. Murugan, S. Ramakrishna, Bioresorbable composite bone paste using polysaccharide-based nano-hydroxyapatite, Biomaterials, 2004, 25, 3829–3835.
- S. V. Dorozhkin, Multiphasic calcium orthophosphate (CaPO4) bioceramics and their biomedical applications, Ceram Int, 2016, 42, 6529–6554.
- I. Denry, L. T. Kuhn, Design, and characterization of calcium phosphate ceramic scaffolds for bone tissue engineering, Dent Mater, 2016, 32, 43–53.
- C. Schwartz, M. Vautrin, Phosphocalcium ceramics are efficient in the management of severe acetabular loss in revision hip arthroplasties. A 22 cases long-term follow-up study, Eur J Orthop Surg Traumatol, 2015, 25, 227–232.
- J. M. Bouler, P. Pilet, O. Gauthier, E. Verron, Biphasic calcium phosphate ceramics for bone reconstruction: A review of biological response, Acta Biomater, 2017, 53, 1–12.
- M. Descamps, O. Richart, P. Hardouin, J. C. Hornez, A. Leriche, Synthesis of macroporous β-tricalcium phosphate with controlled porous architectural, Ceram Int, 2008, 34, 1131–1137.
- P. Ducheyne, Q. Qiu, Bioactive ceramics: the effect of surface reactivity on bone formation and bone cell function, Biomaterials, 1999, 20, 2287-2303.
- I. S. Neira, Y. V. Kolen’ko, O. I. Lebedev, G. Van Tendeloo, H. S. Gupta, F. Guitián, M. Yoshimura, An Effective Morphology Control of Hydroxyapatite Crystals via Hydrothermal Synthesis, Cryst Growth Des, 2009, 9, 466–474.
- M. Okada, T. Matsumoto, Synthesis and modification of apatite nanoparticles for use in dental and medical applications, Jpn Dent Sci Rev, 2015, 51, 85–95.
- S. Rujitanapanich, P. Kumpapan, P. Wanjanoi, Synthesis of Hydroxyapatite from Oyster Shell via Precipitation, Energy Procedia, 2014, 56, 112–117.
- G. J. Owens, R. K. Singh, F. Foroutan, M. Alqaysi, C.-M. Han, C. Mahapatra, H.-W. Kim, J. C. Knowles, Sol–gel-based materials for biomedical applications, Prog Mater Sci, 2016, 77, 1–79.
- M. H. Fathi, A. Hanifi, Evaluation and characterization of nanostructure hydroxyapatite powder prepared by simple sol-gel method, Mater Lett, 2007, 61, 3978–3983.
- M. H. Fathi, A. Hanifi, V. Mortazavi, Preparation and bioactivity evaluation of bone-like hydroxyapatite nanopowder, J Mater Process Technol, 2008, 202, 536–542.
- R. Narayanan, S. K. Seshadri, T. Y. Kwon, K. H. Kim, Calcium phosphate-based coatings on titanium and its alloys, J Biomed Mater Res B Appl Biomater, 2008, 85, 279–299.
- M. Yoshimura, H. Suda, K. Okamoto, K. Ioku, Hydrothermal synthesis of biocompatible whiskers, J Mater Sci, 1994, 29, 3399–3402.
- M. Sadat-Shojai, M.-T. Khorasani, E. Dinpanah-Khoshdargi, A. Jamshidi, Synthesis methods for nanosized hydroxyapatite with diverse structures, Acta Biomater, 2013, 9, 7591–7621.
- A. K. Nayak, Hydroxyapatite Synthesis Methodologies: An Overview, Int. J. ChemTech Res, 2010, 2, 903-907.
- K. Ioku, M. Kamitakahara, Hydroxyapatite Ceramics for Medical Application Prepared By Hydrothermal Method, Phosphorus Res Bull, 2009, 23, 25–30.
- M. Sadat-Shojai, Preparation of hydroxyapatite nanoparticles: comparison between hydrothermal and solvo-treatment processes and colloidal stability of produced nanoparticles in a dilute experimental dental adhesive, J Iran Chem Soc, 2009, 6, 386–392.
- T. Amna, Valorization of Bone Waste of Saudi Arabia by Synthesizing Hydroxyapatite, Appl Biochem Biotechnol, 2018, 186, 779–788.
- H. Chen, B. H. Clarkson, K. Sun, J. F. Mansfield, Self-assembly of synthetic hydroxyapatite nanorods into an enamel prism-like structure,
J Colloid Interface Sci, 2005, 288, 97–103.
- K. Lin, J. Chang, R. Cheng, M. Ruan, Hydrothermal microemulsion synthesis of stoichiometric single-crystal hydroxyapatite nanorods with mono-dispersion and narrow-size distribution, Mater Lett, 2007, 61, 1683–1687.
- Y. Sun, G. Guo, Z. Wang, H. Guo, Synthesis of single-crystal HAP nanorods, Ceram Int, 2006, 32, 951–954.
- M. S. Arsad, P. M. Lee, L. K. Hung, Synthesis and characterization of hydroxyapatite nanoparticles and β-TCP particles, In, 2nd international conference on the biotechnology and food science, 2011, 7, 184–188.
- B. Chen, Z. Zhang, J. Zhang, Q. Lin, D. Jiang, Fabrication and mechanical properties of β-TCP pieces by gel-casting method, Mater Sci Eng C, 2008, 28, 1052–1056.
- G. Felício-Fernandes, M. Laranjeira, Calcium phosphate biomaterials from marine algae. Hydrothermal synthesis and characterization, Quimica Nova, 2000, 23, 441–446.
- E. Landi, A. Tampieri, G. Celotti, S. Sprio, Densification behavior and mechanisms of synthetic hydroxyapatites, J Eur Ceram Soc, 2000, 20, 2377–2387.
- K. Ozeki, Y. Fukui, H. Aoki, Influence of the calcium phosphate content of the target on the phase composition and deposition rate of sputtered films, Appl Surf Sci, 2007, 253, 5040–5044.
- S. Kongsri, K. Janpradit, K. Buapa, S. Techawongstien, S. Chanthai, Nanocrystalline hydroxyapatite from fish scale waste: Preparation, characterization, and application for selenium adsorption in aqueous solution, Chem Eng J, 2013, 215–216, 522–532.
- M. Markovic, B. O. Fowler, M. S. Tung, Preparation and comprehensive characterization of a calcium hydroxyapatite reference material, J Res Natl Inst Stand Technol, 2004, 109, 553-568.
- S. Raynaud, E. Champion, D. Bernache-Assollant, P. Thomas, Calcium phosphate apatites with variable Ca/P atomic ratio I. Synthesis, characterization and thermal stability of powders, Biomaterials, 2002, 23, 1065–1072.
- K. Ishikawa, P. Ducheyne, S. Radin, Determination of the Ca/P ratio in calcium-deficient hydroxyapatite using X-ray diffraction analysis, J Mater Sci Mater Med, 1993, 4, 165–168.
- R. Z. LeGeros, Calcium phosphates in oral biology and medicine, Karger: Basel ; New York, 1991.
- W. Suchanek, M. Yoshimura, Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants, J Mater Res, 1998, 13, 94–117.
- M. Valletregi, Synthesis, and characterization of calcium-deficient apatite, Solid State Ion, 1997, 101–103, 1279–1285.
DOI: http://dx.doi.org/10.13171/mjc10402004221251nr
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
