Experimental and theoretical study of adsorption of L-cysteine on the passivation and corrosion inhibition of aluminum alloy in alkaline environment
Abstract
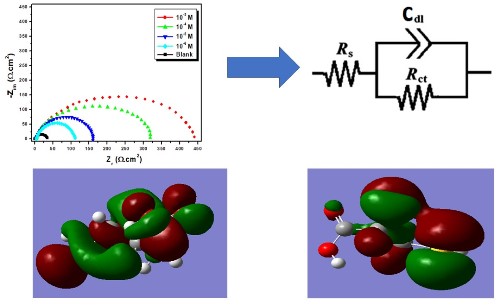
L-Cysteine (L-Cyst) was tested as a corrosion inhibitor of 3003 aluminum alloy (AA3003) in Na2CO3 0.1M + NaCl 1M solution. The inhibition action of this inhibitor was studied by using electrochemical impedance spectroscopy (EIS), potentiodynamic polarization (PDP), and cyclic voltammetry (CV) techniques. The temperature effect on the inhibition efficiency was studied in the range from 298 to 328K, and the values of activation thermodynamic parameters were calculated and explained. Experiment results show that inhibition efficiency increases with the increase of L-Cysteine concentration, and its inhibition efficiency reaches 92% at 10−3M. The inhibition efficiency decreases slightly with the elevation of a corrosive medium temperature. Analysis of the polarization curves revealed that L-Cyst is considered a mixed inhibitor. The results from electrochemical measurements and theoretical calculations are in good agreement. The morphological changes of the surface during corrosion, in the presence and absence of the inhibitor, were studied by scanning electron microscopy (SEM).
Full Text:
PDFReferences
- M. Dabalà, E. Ramous, M. Magrini, Corrosion resistance of cerium‐based chemical conversion coatings on AA5083 aluminium alloy, Mater. Corros., 2004, 55, 381–386.
- A. Y. Musa, A. A. H. Kadhum, A. B. Mohamad, M. S. Takriff, A. R. Daud, S. K. Kamarudin, On the inhibition of mild steel corrosion by 4-amino-5-phenyl-4H-1, 2, 4-trizole-3-thiol, Corros. Sci., 2010, 52, 526–533.
- R. Rosliza, W. B. W. Nik, S. Izman, Y. Prawoto, Anti-corrosive properties of natural honey on Al–Mg–Si alloy in seawater, Curr. Appl. Phys., 2010, 10, 923–929.
- J. A. Hill, T. Markley, M. Forsyth, P. C. Howlett, B. R. W. Hinton, Corrosion inhibition of 7000 series aluminium alloys with cerium diphenyl phosphate, J. Alloy. Compd., 2011, 509, 1683–1690.
- R. Rosliza, W. B. W. Nik, H. B. Senin, The effect of inhibitor on the corrosion of aluminum alloys in acidic solutions, Mater. Chem. Phys., 2008, 107, 281–288.
- S. Gudić, L. Vrsalović, M. Kliškić, I. Jerković, A. Radonić, M. Zekić, Corrosion Inhibition of AA 5052 Aluminium Alloy in NaCl Solution by Different Types of Honey, Int. J. Electrochem. Sci., 2016, 11, 998–1011.
- E. E. Oguzie, G. N. Onuoha, E. N. Ejike, Effect of Gongronema latifolium extract on aluminium corrosion in acidic and alkaline media, Pigment Resin Technol., 2007, 36, 44–49.
A. M. Al-Turkustani, S. T. Arab, R. H. Al-Dahiri, Aloe Plant Extract as Environmentally Friendly Inhibitor on the Corrosion of Aluminum in Hydrochloric Acid in Absence and Presence of Iodide Ions, Mod. Appl. Sci., 2010, 4, 105–124.
- J. Li, B. Hurley, R. Buchheit, Micro electrochemical Characterization of the Effect of Rare Earth Inhibitors on the Localized Corrosion of AA2024-T3, J. Electrochem. Soc., 2015, 162, C563–C571.
- G. O. Ilevbare, J. R. Scully, Mass-Transport-Limited Oxygen Reduction Reaction on AA2024-T3 and Selected Intermetallic Compounds in Chromate-Containing Solutions, Corrosion, 2001, 57, 134–152.
- X. Li, S. Deng, Inhibition effect of Dendrocalamus brandisii leaves extract on Aluminum in HCl, H3PO4 solutions, Corros. Sci., 2012, 65, 299–308.
- S. M. A. Hosseini, A. Azimi, The inhibition of mild steel corrosion in acidic medium by 1-methyl-3-pyridin-2-yl-thiourea, Corros. Sci., 2009, 51, 728–732.
- C. M. Goulart, A. Esteves-Souza, C. A. Martinez-Huitle, C. J. F. Rodrigues, M. A. M. Maciel, A. chevarria, Experimental and theoretical evaluation of semicarbazones and thiosemicarbazones as organic corrosion inhibitors, Corros. Sci., 2013, 67, 281–291.
- W. Li, Q. He, C. Pei, B. Hou, Experimental and theoretical investigation of the adsorption behaviour of new triazole derivatives as inhibitors for mild steel corrosion in acid media, Electrochim. Acta, 2007, 52, 6386–6394.
- F. Bentiss, M. Lagrenee, M. Traisnel, J. C. Hornez, The Corrosion Inhibition of Mild Steel in Acidic Media by a New Triazole Derivative, Corros. Sci., 1999, 41, 789–803.
- A. M. Fekry, R. R. Mohamed, Acetyl thiourea chitosan as an eco-friendly inhibitor for mild steel in sulphuric acid medium, Electrochim. Acta, 2010, 55, 1933–1939.
- K. F. Khaled, The inhibition of benzimidazole derivatives on corrosion of iron in 1 M HCl solutions, Electrochim. Acta, 2003, 48, 2493–2503.
- K. F. Khaled, Experimental, density function theory calculations and molecular dynamics simulations to investigate the adsorption of some thiourea derivatives on iron surface in nitric acid solutions, Appl. Surf. Sci., 2010, 256, 6753–6763.
- M. Mehdipour, B. Ramezanzadeh, S. Y. Arman, Electrochemical noise investigation of Aloe plant extract as green inhibitor on the corrosion of stainless steel in 1 M H2SO4, J. Ind. Eng. Chem., 2015, 21, 318–327.
- A. M. Abdel-Gaber, B. A. Abd-El-Nabey, I. M. Sidahmed, A. M. El-Zayady, M. Saadawy, Inhibitive action of some plant extracts on the corrosion of steel in acidic media, Corros. Sci., 2006, 48, 2765–2779.
- E. Azzouyahar, L. Bazzi1, M. Essahli, M. Belkhaouda, A. Lamiri, Nigella Sativa Oil as Green Corrosion Inhibitor for Aluminum in Na2CO3 Solution, Journal of applicable chemistry, 2014, 4, 1602-1612.
- K. Barouni, L. Bazzi, R. Salghi, M. Mihit, B. Hammouti, A. Albourine, S. El Issami, Some amino acids as corrosion inhibitors for copper in nitric acid solution, Materials Letters, 2008, 62, 3325-3327.
- M. Chadili, M. M. Rguiti, B. El Ibrahimi, R. Oukhrib, A. Jmiai, M. Beelkhaouda, L. Bammou, M. Hilali, L. Bazzi, Corrosion Inhibition of 3003 Aluminum Alloy in Molar Hydrochloric Acid Solution by Olive Oil Mill Liquid By-Product, International Journal of Corrosion, 2021. https://doi.org/10.1155/2021/6662395.
- R. Salghi, L. Bazzi, B. Hammouti, A. Bouchtart, S. Kertit, Z. A. Ait addi, Z. El alami, Étude électrochimique de l'inhibition de la corrosion de l'alliage d'aluminium 3003 en milieu bicarbonate par les composés triazolique, Ann, Chim, Sci. Mat., 2000, 25, 187-200.
- K. Shalabi, Y. M. Abdallah, A. S. Fouda, Corrosion inhibition of Aluminum in 0.5 M HCl solutions containing phenyl sulfonyl acetophenone azo derivatives, Res. Chem. Intermed., 2014, 41, 4687–4711.
- D. Becke, A new mixing of Hartree–Fock and local density‐functional theories, J. Chem. Phy., 1993, 98, 1372.
- C. Lee, W. Yang, R. G. Parr, Development of the Colic-Salvetti correlation-energy formula into a functional of the electron density, Phy. Rev. B, 1988, 37, 785.
- D. Ramirez, R. Vera, H. H. Gomez, C. Conajahua, Evaluation of protecting properties of epoxy coatings on Navy steel by Electrochemical Impedance Spectroscopy, J. Chil. Chem. Soc., 2005, 50, 489-494.
- L. Bazzi, S. Kertit, M. Hamdani, Effet de l'addition d'oxo-anions sur le comportement à la corrosion de l'alliage d'aluminium 6063 en milieu hydrogénocarbonate, J. Chim. Phys., 1995, 92, 1612-1628.
- M. Belkhaouda, L. Bazzi, A. Benlhachemi, R. Salghi, B. Hammouti, A. Ihlal, Influence of the heat treatment on the corrosion behaviour of 3003 aluminium alloy in alkaline medium, Phys. Chem. News, 2009, 48, 81-88.
- L. Bazzi, S. Kertit, M. Hamdani, Effet de l'addition d'ions chromates sur le comportement à la corrosion d'un alliage d'aluminium 6063 en milieu neutre, pollué par les ions cuivriques, J. Chim. Phys., 1997, 94, 93-104.
- R. Ambat, E. S. Dwarakadasa, The influence of pH on the corrosion of medium strength aerospace alloys 8090, 2091 and 2014, Corros. Sci., 1992, 33, 681-690.
- K. R. Ansari, D. K. Yadav, E. E. Ebenso, M. A. Quraishi, Novel and Effective Pyridyl Substituted 1,2,4-Triazole as Corrosion Inhibitor for Mild Steel in Acid Solution, Int. J. Electrochem. Sci., 2012, 7, 4780-4799.
- K. Toumiat, Y. El Aoufir, H. Lgaz, R. Salghi, S. Jodeh, M. Zougagh, Monte Carlo Simulation and Electrochemical Assessment of Inhibitive Behavior of Pyrazole Derivative On Mild Steel in HCl Medium, Res. J. Pharm. Biol. Chem. Sci., 2016, 7, 1209–1217.
- S. Kharacha, A. Batah, M. Belkhaouda, L. Bazzi, L. Bammou, R. Salghi, O. Jbara, A. Tara, Effect of amino acid on the passivation, corrosion and inhibition behavior of aluminum alloy in alkaline medium, Mor. J. Chem., 2018, 6, 294-306.
- O. Krim, S. Jodeh, M. Messali, B. Hammouti, A. Elidrissi, K. Khaled, Synthesis, Characterization and Corrosion Protection Properties of Imidazole Derivatives on Mild Steel in 1.0 M HCl, Port Electrochimica Acta, 2016, 34, 213–229.
- B. El Makrini, H. Lgaz, K. Toumiat, R. Salghi, S. Jodeh, G. Hanbali, Adsorption and Corrosion-Inhibiting Effect of 5-Benzyloxy-6-methoxyindole on Mild Steel Surface in Hydrochloric Acid Solution: Electrochemical and Monte Carlo simulation studies, Res. J. Pharm. Biol. Chem. Sci., 2016, 7, 2277–2285.
- B. El Makrini, K. Toumiat, H. Lgaz, R. Salghi, S. Jodeh, G. Hanbali, Corrosion protection of mild steel in hydrochloric acid solutions in presence of 5-(Benzyloxy) indole – Monte Carlo simulation, weight loss and electrochemical studies, Res. J. Pharm. Biol. Chem. Sci., 2016, 7, 2286–2294.
- N. O. Eddy, H. Momoh-Yahaya, E. E. Oguzie, Theoretical and experimental studies on the corrosion inhibition potentials of some purines for Aluminum in 0.1 M HCl, J Adv Res., 2015, 6, 203-217.
- D. Zhang, Y. Tang, S. Qi, D. Dong, H. Cang,
G. Lu, The inhibition performance of long-chain alkyl-substituted benzimidazole derivatives for corrosion of mild steel in HCl, Corros Sci., 2016, 102, 517–522.
- K. F. Khaled, Studies of iron corrosion inhibition using chemical, electrochemical and computer simulation techniques, Electrochimica Acta, 2010, 55, 6523–6532.
- P. Muthukrishnan, P. Prakash, B. Jeyaprabha, K. Shankar, Stigmasterol extracted from Ficus hispida leaves as a green inhibitor for the mild steel corrosion in 1 M HCl solution, Arab J Chem., 2019, 12, 3345-3356.
- M. Lebrini, M. Lagrenée, H. Vezin, M. Traisnel, F. Bentiss, Experimental and theoretical study for corrosion inhibition of mild steel in normal hydrochloric acid solution by some new macrocyclic polyether compounds, Corros. Sci., 2007, 49, 2254–2269.
- I. B. Obot, N. O. Obi-Egbedi, N. W. Odozi, Acenaphtho [1,2-b] quinoxaline as a novel corrosion inhibitor for mild steel in 0.5 M H2SO4, Corros. Sci., 2010, 52, 923–926.
- N. Guan, M. L. Xueming, L. Fei, Synergistic inhibition between o-phenanthroline and chloride ion on cold-rolled steel corrosion in phosphoric acid, Chem. Phys., 2004, 86, 59-68.
- F. Bentiss, M. Lebrini, M. Lagrenée, Thermodynamic characterization of metal dissolution and inhibitor adsorption processes in mild steel/ 2,5-bis(n-thienyl)-1,3,4-thiadiazoles/ hydrochloric acid system, Corros. Sci., 2005, 47, 2915-2931.
- X. Li, S. Deng, H. Fu, Triazolyl blue tetrazolium bromide as a novel corrosion inhibitor for steel in HCl and H2SO4 solutions, Corros. Sci., 2011, 53, 302-309.
- F. Mansfeld, M. W. Kending, S. Tsai, Recording and Analysis of AC Impedance Data for Corrosion Studies, Corros., 1981, 37, 301-307.
- F. Beck, U. A. Kruger, EIS of cathodically deposited wet paint films prior to the stoving process, Electrochim. Acta, 1996, 41, 1083-1092.
- Z. El Adnani, M. Mcharfi, M. Sfaira, A. T. Benjelloun, M. Benzakour, M. EbnTouhami, B. Hammouti, M. Taleb, Investigation of Newly Pyridazine Derivatives as Corrosion Inhibitors in Molar Hydrochloric Acid. Part III: Computational Calculations, Int. J. Electrochem. Sci., 2012, 7, 3982–3996.
- R. G. Pearson, Hard and soft acids and bases-the evolution of a chemical concept, Coordination Chemistry Reviews, 1990, 100, 403–425.
- A. Kokalj, N. Kovačević, On the consistent use of electrophilicity index and HSAB-based electron transfer and its associated change of energy parameters, Chemical Physics Letters, 2011, 507, 181–184.
- S. Kharacha, I. Bazzi, M. Belkhaouda, L. Bammou, A. Batah, L. Bazzi, R. Salghi, M. Hilali, Effect of L-methionine on the passivation, corrosion and inhibition behavior of tin in alkaline medium, Appl. J. Envir. Eng. Sci., 2018, 4, 339-413.
DOI: http://dx.doi.org/10.13171/mjc02103041563sk
Refbacks
- There are currently no refbacks.
Copyright (c) 2021 Mediterranean Journal of Chemistry
