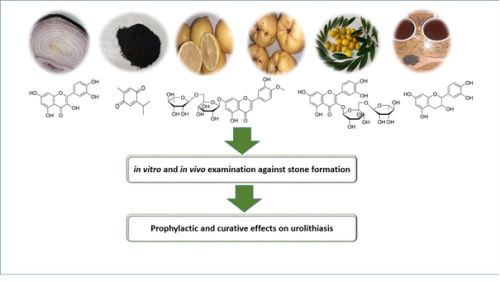
Phytomolecules investigated for the prevention and treatment of urinary stones
Abstract
Urolithiasis is a recurrent pathology manifested by the stone formation in the urinary system; it has long been treated in traditional medicine by plant remedies. Several studies have provided the efficacy of medicinal plants as well as their chemical compounds against stone formation. The present work aims to summarize the antiurolithiatic effect of phytochemicals, including quercetin, rutin, catechin, diosmin, and thymoquinone. In this context, various databases, including PubMed, Science Direct, Scopus, and google scholar, were searched using keywords like antiurolithiatic bioactive molecules and chemistry of phytomolecules.
The results confirmed that phytochemicals, including particularly flavonoid molecules, could be effective against lithogenesis via different strategies such as decreasing the binding between crystals and cells, decreasing the growth of crystals, and increasing magnesium level. Nevertheless, more studies are required, such as determination of toxicity and clinical studies. This review may help researchers achieve more results about the mechanism and the side effects of phytochemicals administration.
Full Text:
PDFReferences
- W. F. McNutt, Chapter VII: Vesical Calculi (Cystolithiasis), in: Diseases of the kidneys and bladder: a text-book for students of medicine, IV: Diseases of the Bladder, J. B. Lippincott Company, Philadelphia, 1893, 185-186.
- D. J. Kok, S. R. Khan, Calcium oxalate nephrolithiasis, a free or fixed particle disease, Kidney Int., 1994, 46, 847–854.
- G. Farell, E. Huang, S. Y. Kim, R. Horstkorte, J. C. Lieske, Modulation of proliferating renal epithelial cell affinity for calcium oxalate monohydrate crystals, J Am Soc Nephrol., 2004, 15, 3052–3062.
- A. K. Shukla, S. Shukla, A. Garg, S. Garg, A review on the anti-urolithiatic activity of herbal folk plants, Asian Journal of Biomaterial Research, 2017, 3, 1-11.
- S. K. Mekap, S. Mishra, S. Sahoo, P. K. Panda, Antiurolithiatic activity of Crataeva magna Lour bark, Indian Journal of Natural Products and Resources, 2011, 1, 28-33.
- A. L. Schwaderer, A. J. Wolfe, The association between bacteria and urinary stones, Ann Transl Med., 2017, 5, 32.
- V. Romero, H. Akpinar, D. G. Assimos, Kidney Stones: A Global Picture of Prevalence, Incidence, and Associated Risk Factors Kidney Stones: A Global Perspective, Reviews in urology, 2010, 12, 86–96.
- S. Gindi, T. Methra, B. R. Chandu, Antiurolithiatic and in vitro anti-oxidant activity of leaves of Ageratum conyzoides in rat, World J. Pharm., 2013, 2, 636–649.
- K. Mikawlrawng, S. Kumar, Current scenario of urolithiasis and the use of medicinal plants as antiurolithiatic agents in Manipur (North East India): A Review, International Journal of Herbal Medicine, 2014, 2, 1-12.
- N. L. Miller, J. E. Lingeman, Management of kidney stones, Br. Med. J., 2007, 334, 468-472.
- R. P. Terlecki, J. A. Triest, A contemporary evaluation of the auditory hazard of extracorporeal shock wave lithotripsy, Urology, 2007, 70, 898-899.
- J. R. S. Tabuti, K. A. Lye, S. S. Dhillion, Traditional herbal drugs of Bulamogi, Uganda: plants, use and administration, J. Ethnopharmacol., 2003, 88, 19-44.
- S. Lipismita, K. P. Ashok, M. Chinmoy, Nutritional strategies to prevent urolithiasis in animals, Veterinary World, 2011, 4, 142-144.
- M. Birjees, M. Ahmad, M. Zafar, S. Nawaz, S. Jehanzeb, F. Ullah, W. Zaman, Traditional knowledge of wild medicinal plants used by the inhabitants of Garam Chashma valley, district Chitral, Pakistan, Acta Ecologica Sinica, 2021. https://doi.org/10.1016/j.chnaes.2020.12.006.
- F. Ulla, A. Ayaz, S. Saqib, W. Zaman, M. A. Butt, A. Ullah, Silene conoidea L.: A review on its systematic, ethnobotany and phytochemical profile, Plant Science Today, 2019, 6, 373-382.
- W. Zaman, M. Ahmad, M. Zafar, H. Amina, F. Ullah, S. Bahadur, A. Ayaz, S. Saqib, N. Begum, S. Jahan, The quest for some novel antifertility herbals used as male contraceptives in district Shangla, Pakistan. Acta Ecologica Sinica, 2020, 40, 102–112.
- M. Ghourri, L. Zidane, A. Douira, Catalogue des plantes médicinales utilisées dans le traitement de la lithiase rénale dans la province de Tan-Tan (Maroc saharien), Int. J. Biol. Chem. Sci., 2013, 7, 1688-1700.
- K. Agarwal, R. Varma, Ethnobotanical study of antilithic plants of Bhopal district, Journal of Ethnopharmacology, 2015, 174, 17-24.
- K. Nemeth, M. K. Piskuła, Food content, processing, absorption and metabolism of onion flavonoids, Crit. Rev. Food Sci. Nutr., 2007, 47, 397–409.
- W. Zeng, S. Y. Wang, Oxygen radical absorbing capacity of phe¬nolics in blueberries, cranberries, chokenberries and lingonber¬ries, J. Agric. Food Chem., 2003, 51, 502–509.
- N. Berardini, R. Fezer, J. Conrad, U. Beifuss, R. Carle, A. Schieber, Screening of mango (Mangifera indica L.) cultivars for their contents of flavonol O- and xanthone C-glycosides, anthocya¬nins and pectin, J. Agric. Food Chem., 2005, 53, 1563-1570.
- H. Liu, L. Zhang, S. P. Lu, Evaluation of antioxidant and immunity activities of quercetin in isoproterenol-treated rats, Molecules, 2012, 17, 4281-4291.
- M. Bule, A. Abdurahman, S. Nikfar, M. Abdollahi, M. Amini, Antidiabetic effect of quercetin: A systematic review and meta-analysis of animal studies, Food Chem. Toxicol., 2019, 125, 494-502.
- H. K. Park, B. C. Jeong, M. K. Sung, Reduction of oxidative stress in cultured renal tubular cells and preventive effects on renal stone formation by the bioflavonoid quercetin, J. Urol., 2008, 179, 1620–1626.
- W. Zhu, Y. F. Xu, Y. Feng, B. Peng, J. P. Che, M. Liu, J. H Zheng, Prophylactic effects of quercetin and hyperoside in a calcium oxalate stone-forming rat model, Urolithiasis, 2014, 42, 519–526.
- B. M. Dinnimath, S. S. Jalalpure, U. K. Patil, Antiurolithiatic activity of natural constituents isolated from Aerva lanata, Journal of Ayurveda and Integrative Medicine, 2017, 8, 226–232.
- A. V. A. David, R. Arulmoli, S. Parasuraman, Overviews of biological importance of quercetin: A bioactive flavonoid, Phcog. Rev., 2016, 10,
-89.
- L. S. Chua, A review on plant-based rutin extraction methods and its pharmacological activities, Journal of Ethnopharmacology, 2013, 150, 805–817.
- G. A. C. Ribeiroa, C. Q. Rochaa, W. B. Veloso, R. N. Fernandesa, I. S. Silvab, A. A. Tanaka, Determination of the catechin contents of bioactive plant extracts using disposable screen-printed carbon electrodes in a batch injection analysis (BIA) system, Microchemical Journal, 2019, 146, 1249–1254.
- P. V. Gadkari, M. Balaraman, Catechins: Sources, extraction and encapsulation: A review, food and bioproducts processing, 2015, 93,
–138.
- E. Cione, C. La Torre, R. Cannataro, M. C. Caroleo, P. Plastina, L. Gallelli, Quercetin, Epigallocatechin Gallate, Curcumin, and Resveratrol: From Dietary Sources to Human MicroRNA Modulation, Molecules, 2020, 25, 63.
- J. Taborsky, M. Kunt, P. Kloucek, J. Lachman, V. Zeleny, L. Kokoska, Identification of potential sources of thymoquinone and related compounds in Asteraceae, Cupressaceae, Lamiaceae, and Ranunculaceae families, Open Chem., 2012, 10, 1899-1906.
- E. A. Cladera, A. Nadal-Casellas, Y. Gómez-Pérez, I. Gomila, R.M. Prieto, A.M. Proenza, I. Lladó, Phytotherapy in a rat model of hyperoxaluriahe antioxidant effects of quercetin involve serum paraoxonase 1 activation, Exp. Biol. Med., 2011, 236, 1133–1138.
- S. Sharma, A. Ali, J. Ali, J. K. Sahni, S. Baboota, Rutin: therapeutic potential and recent advances in drug delivery, Expert Opinion on Investigational Drugs, 2013, 22, 1063-1079.
- A. Kanashiro, D. C. Andrade, L. M. Kabeya, Modulatory effects of rutin on biochemical and hematological parameters in hypercholesterolemic Golden Syrian hamsters, An. Acad. Bras. Cienc., 2009, 81, 67–72.
- J. P. Lin, J. S. Yang, J. J. Lin, K. C. Lai, H. F. Lu, C. Y. Ma, C. Wu, K. C. Wu, F. S. Chueh, W. G. Wood, J. G. Chung, Rutin inhibits human leukemia tumor growth in a murine xenograft model in vivo, Environmental Toxicology, 2012, 27, 480-484.
- N. T. Niture, A. A. Ansari, S. R. Naik, Anti-hyperglycemic activity of rutin in streptozotocin-induced diabetic rats: an effect mediated through cytokines, antioxidants and lipid biomarkers, Indian Journal of Experimental Biology, 2014, 52, 720-727.
- S. Wang, C. Wang, L. Gao, H. Cai, Y. Zhou, Y. Yang, C. Xu, W. Ding, J. Chen, I. Muhammad, X. Chen, X. He, D. Liu, Y. Li, Rutin inhibits Streptococcus suis biofilm formation by affecting CPS biosynthesis, Frontiers in Pharmacology, 2017, 8, 1-12.
- F.M. Kandemir, M. Ozkaraca, B. A Yildirim, B. Hanedan, A. Kirbas, K. Kilic, E. Aktas, F. Benzer, Rutin attenuates gentamicin-induced renal damage by reducing oxidative stress, inflammation, apoptosis, and autophagy in rats, Renal Failure, 2015, 37, 518-525.
- W. Zhai, J. Zheng, X. Yao, B. Peng, M. Liu, J. Huang, G. Wang, Y. Xu, Catechin prevents the calcium oxalate monohydrate induced renal calcium crystallization in NRK-52E cells and the ethylene glycol induced renal stone formation in rat, BMC Complement. Altern. Med., 2013, 13, 1-11.
- J. Ghodasara, A. Pawar, C. Deshmukh, Inhibitory effect of rutin and curcumin on experimentally-induced calcium oxalate urolithiasis in rats, Pharmacognosy Res., 2011, 2, 388-392.
- K. Kaihatsu, M. Yamabe, Y. Ebara, Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters, Molecules, 2018, 23, 2475.
- Y. Shirakami, M. Shimizu, Possible Mechanisms of Green Tea and Its Constituents against Cancer, Molecules, 2018, 23, 2284.
- S. Y. Lin, L. Kang, C. Z. Wang, H. H. Huang, T. L. Cheng, H. T. Huang, M. J. Lee, Y. S. Lin, M. L. Ho, G. J. Wang, Epigallocatechin-3-Gallate (EGCG) Enhances Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells, Molecules, 2018, 23, 3221.
- S. T. Chen, L. Kang, C. Z. Wang, P. J. Huang, H. T. Huang, S. Y. Lin, S. H. Chou, C. C. Lu, P. C. Shen, Y. S. Lin, Epigallocatechin-3-Gallate Decreases Osteoclastogenesis via Modulation of RANKL and Osteoprotegrin, Molecules, 2019, 24, 156.
- G. H. Bao, J. Xu, F. L. Hu, X. C. Wan, S. X. Deng, J. Barasch, EGCG inhibit chemical reactivity of iron through forming a Ngal-EGCG-iron complex, Biometals, 2013, 26, 1041–50.
- F. Grases, R. M. Prieto, R. A. Fernandez-Cabot, A. Costa-Bauzá, F. Tur, J. J. Torres, Effects of polyphenols from grape seeds on renal lithiasis, Oxid. Med. Cell. Longev., 2015, 1-6.
- X. Li, G. Wu, P. Shang, Anti-nephrolithic potential of catechin in melamine-related urolithiasis via the inhibition of ROS, apoptosis, phospho-p38 and osteopontin in male Sprague-Dawley rats, Free Radical Research, 2015, 49,1249-1258.
- R. Kanlaya, N. Singhto, V. Thongboonkerd, EGCG decreases binding of calcium oxalate monohydrate crystals onto renal tubular cells via decreased surface expression of alpha-enolase, J. Biol. Inorg. Chem., 2016, 21, 339–346.
- B. C. Jeong, B. S. Kim, J. I. Kim, H. H. Kim, Effects of green tea on urinary stone formation in vivo and in vitro study, J. Endourol., 2006, 20, 356–361.
- A. Bogucka-Kocka, M. Woźniak, M. Feldo, J. Kocki, K. Szewczyk, Diosmin-Isolation Techniques, Determination in Plant Material and Pharmaceutical Formulations, and Clinical Use, Nat. Prod. Commun., 2013, 8.
- M. U. Rehman, M. Tahir, A. Quaiyoom Khan, R. Khan, A. Lateef, O. O. Hamiza, F. Ali, S. Sultana, Diosmin protects against trichloroethylene-induced renal injury in Wistar ratslausible role of p53, Bax and caspases, Br. J. Nutr., 2013, 110, 699–710.
- A. Noorafshan, S. Karbalay-Doust, F. Karimi, Diosmin reduces calcium oxalate deposition and tissue degeneration in nephrolithiasis in rats: A stereological study, Korean J. Urol., 2013, 54, 252–257.
- V. V. Prabhu, D. Sathyamurthy, A. Ramasamy, S. Das, M. Anuradha, S. Pachiappan, Evaluation of protective effects of diosmin (a citrus flavonoid) in chemical-induced urolithiasis in experimental rats, Pharm. Biol., 2016, 54, 1513–1521.
- A. Ahmad, R. K. Mishra, A. Vyawahare, A. Kumar, M. U. Rehman, W. Qamar, R. Khan, Thymoquinone (2-Isopropyl-5-methyl-1, 4-benzoquinone) as a chemopreventive/anticancer agent: Chemistry and biological effects, Saudi Pharm. J., 2019, 27, 1113–1126.
- M. Hajzadeh, N. Mohammadian, Z. Rahmani, F. B. Rassouli, Effect of thymoquinone on ethylene glycol-induced kidney calculi in rats, Urol. J., 2008, 5, 149–155.
- P. Hayatdavoudi, A. Khajavi Rad, Z. Rajaei, M. A. R. Hadjzadeh, Renal injury, nephrolithiasis and Nigella sativa: A mini review, Avicenna J Phytomed., 2016, 6, 1-8.
- X. Tang, J. C. Lieske, Acute and chronic kidney injury in nephrolithiasis, Curr Opin Nephrol Hypertens., 2014, 23, 385–390.
- D. R. Mandavia, M. K. Patel, J. C. Patel, A. P. Anovadiya, S. N. Baxi, C. R. Tripathi, Anti-urolithiatic effect of ethanolic extract of Pedalium murex linn. fruits on ethylene glycol-induced renal calculi, Urol. J., 2013, 10, 946–952.
DOI: http://dx.doi.org/10.13171/mjc02102271568fee
Refbacks
- There are currently no refbacks.
Copyright (c) 2021 Mediterranean Journal of Chemistry
