Cyclic and differential pulse voltammetry investigations of an iodine contrast product using microelectrode of BDD
Abstract
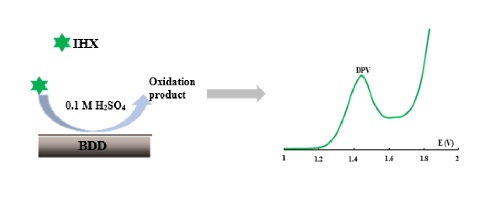
This work deals with the electrochemical behavior and detection of an iodine contrast product using microelectrode. For this, cyclic voltammetry and differential pulse were used for behavior study and detection, respectively. In this work, a boron-doped diamond (BDD) microelectrode was used. Cyclic voltammetry showed that the oxidation peak of iohexol (IHX) appeared at a potential of 1.655 V/ESM in H2SO4 medium (0.1 M). The electrooxidation process of IHX is irreversible, controlled by diffusion combined par adsorption, and can undergo direct and/or indirect oxidation. In addition, using the differential pulse voltammetry technique, the peak oxidation current shows a linear relationship with the IHX concentration between 4 µmol/L and 74.35 µ mol/L. The calculated limit of detection (LOD) and limit of quantification (LOQ) are 1.953 µmol/L and 6.511 µmol/L, respectively. Recovery rates ranging from 95% to 99% in the presence of interfering compounds (inorganic compounds with concentrations 300 times higher than that of IHX). In addition, the DDB successfully recovered the concentration (20 µmol/L) of IHX dissolved with paracetamol concentrations ranging from 31 µmol/L to 149.66 µmol/L. These results suggest that the BDD can be used as a practical sensor to detect and quantify trace amounts of IHX.
Full Text:
PDFReferences
M. Schlüsener, D. Löffler, T. Ternes, List of the relevant PPs, In Projet Européen KNAPPE, deliverable D11, European Commission, 6th Framework Program, 2008, 63.
A. Sadezky, D. Löffler, T. Ternes, Proposal of an environmental indicator and classification system of pharmaceutical product residues for environmental management, In Projet Européen KNAPPE, deliverable D12, European Commission, 6th Framework Program, 2008, 92.
X.-D. Lv, Y.-H. Cui, W.-J. Xue, S.-Q. Yang, J.Y. Li, Z.Q. Liu, Comparison of inert and non-inert cathode in cathode/Fe3þ/ Peroxymonosulfate processes on iohexol degradation, Chemosphere, 2019, 223, 494-503.
J. Jeong, J. Jung, W.J. Cooper, W. Song, Degradation mechanisms and kinetic studies for the treatment of X-ray contrast media compounds by advanced oxidation/ reduction processes, Water Res., 2010, 44, 4391–4398.
F.X. Tian, B. Xu, Y.L. Lin, C.Y. Hu, T.Y. Zhang, N.Y. Gao, Photodegradation kinetics of iopamidol by UV irradiation and enhanced formation of iodinated disinfection by-products in sequential oxidation processes, Water Res., 2014, 58, 198–208.
S.E. Duirk, C. Lindell, C.C. Cornelison, J. Kormos, T.A. Ternes, M. Attene-Ramos, J. Osiol, E.D. Wagner, M.J. Plewa, S.D. Richardson, Formation of toxic iodinated disinfection by-products from compounds used in medical imaging, Environ. Sci. Technol., 2011,45, 6845–6854.
R.L. Oulton, T. Kohn, D.M. Cwiertny, Pharmaceuticals and personal care products in effluent matrices: a survey of transformation and removal during wastewater treatment and implications for wastewater management, J. Environ. Monit., 2010, 12, 1956–1978.
S. Castagnet, H. Blasco, P. Vourc'h P, I. Benz-De-Bretagne, C. Veyrat-Durebex, C. Barbet, A. Alnajjar, B. Ribourtout, M. Buchler, J.-M. Halimi, C. R Andres, Routine determination of GFR in renal transplant recipients by HPLC quantification of plasma iohexol concentrations and com-parison with estimated GFR, J. Clin. Lab. Anal., 2012, 26,376–83.
E. Cavalier, E. Rozet, N. Dubois, C. Charlier, P. Hubert, J.-P. Chapelle, J.-M. Krzesinski, P. Delanaye, performance of iohexol determination in serum and urine by HPLC: validation, risk and uncertainty assessment, Clin. Chim. Acta, 2008, 396, 80–85.
A. Slack, M. Tredger, N. Brown, B. Corcoran, K. Moore, Application of an isocratic methanol-ba-sed HPLC method for the determination of iohexol concentrations and glomerular filtration rate in patients with cirrhosis, Ann. Clin. Biochem., 2014, 51, 80–88.
M.C. Denis, K. Venne, D. Lesiege, M. Francoeur, S. Groleau, M. Guay, J. Cusson, A. Futos, Development and evaluation of a liquid chromatography-mass spectrometry assay and its application for the assessment of renal function, J. Chromatogr. A, 2008, 1189,
– 416.
E. A. McGaw, G.M. Swain, A comparison of boron-doped diamond thin-film and Hg-coated glassy carbon electrodes for anodic stripping voltammetric determination of heavy metal ions in aqueous media, Anal. Chim. Acta, 2006, 575, 180-189.
E.K. Kouadio, O. Kambire, K.S. Koffi, L. Ouattara, Electrochemical oxidation of paracetamol on boron-doped diamond electrode: analytical performance and paracetamol degradation, J. Electrochem. Sci. Eng., 2021, 11, 71-86.
K.M. Koffi, L. Ouattara, Electroanalytical investigation on paracetamol on boron-doped diamond electrode by Voltammetry, Am. J. Anal. Chem., 2019, 10, 562-578.
P. Niedziałkowski, Z. Cebula, N. Malinowska, W. Białobrzeska, M. Sobaszek, M. Ficek, R. Bogda-nowicz, J. Sein Anand, T. Ossowski, Comparison of the paracetamol electrochemical determination using boron-doped diamond electrode and boron-doped carbon nanowalls, Biosens. Bioelectron., 2019, 126, 308-314.
H. Li, K. Zhou, J. Cao, Q. Wei, C.-T. Lin, S.E. Pei, L. Ma, N. Hu, Y. Guo, Z. Deng, Z. Yu, S. Zeng, W. Yang, L. Meng, A novel modification to boron-doped diamond electrode for enhanced, selective detection of dopamine in human serum, Carbon, 2021,171, 16-28.
L. Švorc, M. Haššo, O. Astrakhan, K. Kianičková, D. M. Stanković, P. Otřísal, A progressive electrochemical sensor for food quality control: Reliable determination of theobromine in chocolate products using a miniaturized boron-doped diamond electrode, Microchem. J., 2018, 142, 297-304.
W. Zhang, I. Soutrel, A. Amrane, F. Fourcade, F. Geneste, Electroreductive deiodination of iohexol catalyzed by vitamin B12 and biodegradability investigation, J. Electroanal. Chem., 2021, 897, 115559.
G.R. Salazar-Banda, L.S. Andrade, P.A.P. Nascente, P.S. Pizani, R.C. Rocha-Filho, L.A. Avaca, The changing electrochemical behaviour of boron-doped diamond surfaces with time after cathodic pre-treatments, Electrochim. Acta, 2006, 51, 4612-4619.
A. A. Ensafi, M. M. Abarghoui, B. Rezaei, Simultaneous determination of morphine and codeine using Pt nanoparticles supported on porous silicon flour modified ionic liquid carbon paste electrode, Sens. Actuators B, 2015, 219, 1-9.
A. Murthy, A. Manthiram, which chemicals drive biological effects in wastewater and recycled Water, Electrochim. Acta, 2011, 56, 6078–6083.
E. Wudarska, E. Chrzescijanska, E. Kusmierek and J. Rynkowski, Electrochemical behavior of 2-(p-isobutylphenyl) propionic Acid at Platinum Electrode, Int. J. Electrochem. Sci., 2015, 10, 9433–9442.
A. Masek, E. Chrzescijanska, Effect of UV-A Irradiation and Temperature on the Antioxidant activity of quercetin studied using ABTS, DPPH and electrochemistry methods, Int. J. Electrochem.Sci., 2015, 10, 5276–5290.
N.P. Shetti, D.S. Nayak, S.D. Bukkitgar, Electrooxidation of antihistamine drug methdilazine and its analysis in human urine and blood samples, Cogent Chem., 2016, 2, 1–13.
P. Kissinger, W.H. Heineman, Laboratory Techniques in electroanalytical chemistry, 2nd ed., New York, 1996.
A. Masek, E. Chrzescijanska, M. Zaborski, Electrooxidation of morin hydrate at a Pt electrode studied by cyclic voltammetry, Food Chem., 2014, 148,18–23.
N. Saadati, M.P. Abdullah, Z. Zakaria, S.B.T. Sany, M. Rezayi, H. Hassonizadeh, Limit of detection and limit of quantification development procedures for organochlorine pesticides analysis in water and sediment matrices, Chem. Cent. J., 2013, 7, 63.
K.M. Koffi, L. Ouattara, Differential pulse voltammetric determination of paracetamol on a boron-doped diamond electrode: application to natural tomato and carrot juices, Rev. Ivoir. Sci. Technol., 2019, 34, 44–66.
M. Berté, F.T.A. Appia, I. Sanogo, L. Ouattara, Electrochemical Oxidation of the Paracetamol in its Commercial Formulation on Platinum and Ruthenium Dioxide Electrodes, Int. J. Electrochem. Sci., 2016, 11, 7736–7749.
L. Nyssen, P. Delanaye, C. Le Goff, S. Peeters, É. Cavalier, A simple LC-MS method for the determination of iohexol and iothalamate in serum, using ioversol as an internal standard, Clin. Chim. Acta, 2016, 463, 96–102.
D.J. Schmit, L.J. Carroll, J. H. Eckfeldt, J.C. Seegmillera, Verification of separate measurement procedures where analytical determinations influence the clinical interpretation of GFR: Iohexol quantitation by HPLC and LC-MS/MS, Clin. Biochem., 2019, 67, 16–23.
F.B. Vicente, G. K. Vespa, F. Carrara,
F. Gaspari, S. Haymond, Determination of iohexol in human serum by a semi-automated liquid chromatography-tandem mass spectrometry method, Clin. Biochem., 2015, 48, 679–685.
S.K.V. Houcke, L. Seaux, E. Cavalier, M.M. Speeckaert, E. Dumoulin, E. Lecocq, J.R. Delanghe, Determination of iohexol and iothalamate in serum and urine by capillary electrophoresis, Electro-phoresis J., 2016, 37, 2363-2367.
L. Zhuang, J. Gao, Y. Zeng, F. Yu, B. Zhang, M. Li, H. Derendorf, C. Liu, HPLC method validation for the quantification of lomustine to study pharmacokinetics of thermosensitive liposome-encapsulated lomustine containing iohexol for CT imaging in C6 glioma rats, Eur. J. Drug Metab. Pharmacokinet, 2011, 36, 61–69.
DOI: http://dx.doi.org/10.13171/mjc02109301594lassiné
Refbacks
- There are currently no refbacks.
Copyright (c) 2021 Mediterranean Journal of Chemistry
