Michael addition of active methylene compounds to (Z)-2-arylidenebenzo[b]thiophen-3(2H)-ones
Abstract
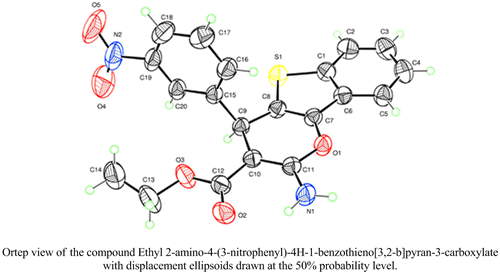
A series of 2-amino-4-aryl-4H-1-benzothieno[3,2-b]pyran-3-carboxylates and 2-amino-4-aryl-3-cyano-4H-1-benzothieno[3,2-b]pyrans has been synthesized through Michael addition of (Z)-2-arylidenebenzo[b]thiophen-3(2H)-ones with active methylene compounds, such as ethyl cyanoacetate and malononitrile. Â The reaction was carried out in ethanol, in the presence of piperidine as a basic catalyst. Â The structures of the prepared compounds were determined by spectroscopic methods: Â 1H-NMR, 13C-NMR and confirmed by single crystal X-Ray diffraction.
Full Text:
PDFReferences
- D. L. Wood, D. Panda, T. R. Wiernicki, L. Wilson, M. A. Jordan, J. P. Singh, Mol. Pharmacol., 1997, 52, 437-444.
- D. Panda, J. P. Singh, L. Wilson, J. Biol. Chem., 1997, 272, 7681–7687.
- M. Kidwai, S. Saxena, M. K. R. Khan, S. S. Thukral, Bioorg. Med. Chem. Lett., 2005,
, 4295–4298.
- D. Kumar, V. Reddy, S. Sharad, U. Dube, S. Kapur, Eur. J. Med. Chem., 2009, 44,
-3809.
- D. C. Mungra, M. P. Patel, D. P. Rajani, R. G. Patel, Eur. J. Med. Chem., 2011, 46,
-4200.
- P. Aragade, V. R. Narayanan, P. Patil, R. Ghule, Pharmtechmedica, 2012, 1, 181-187.
- A. E-G. E. Amr, S. S. Maigali, M. M. Abdulla, Monatsh Chem, 2008, 139, 1409-1415.
- R. M. Schultz, J. D. Papamatheakis, W. A. Stylos, M. A. Chirigos, Cell. Immunology,
, 25, 309-316.
- A.-G. E. Amr, A. M. Mohamed, S. F. Mohamed, N. A. Abdel-Hafez, A. E-F. G. Hammam, Bioorg. Med. Chem., 2006, 14, 5481-5488.
- S. Makarem, A. A. Mohammadi, A. R. Fakhari, Tetrahedron Lett., 2008, 49, 7194-7196.
- K. Auwers and F. Arndt, Ber., 1909, 42, 537-545.
- P. Friendlaender, Monatsh. Chem., 1909, 30, 347-354.
- G. Wagner, B. Eppner, Pharmazie, 1979, 34, 527-530.
- I. W. Still, P.C. Arora, M. S. Chauhan, M-H. Kwan, M. T. Thomas, Can. J. Chem., 1976, 54, 455-470.
- D. H. Wadsworth, M. R. Detty, J. Org. Chem., 1980, 45, 4611-4615.
- T. Yamaguchi, T. Seki, T. Tamaki, K. Ichimura, Bull. Chem. Soc. Jpn., 1992, 65,
-656.
- T. Seki, T. Tamaki, T. Yamaguchi, K. Ichimura, Bull. Chem. Soc. Jpn., 1992, 65, 657-663.
- L. Somogyi, Can. J. Chem., 2001, 79, 1159-1165.
- M. G. Cabiddu, S. Cabiddu, E. Cadoni, S. De Montis, C. Fattuoni, S. Melis, M. Usai, Synthesis, 2002, 875-878.
- T. K. Pradhan, A. De, J. Mortier, Tetrahedron, 2005, 61, 9007-9017.
- A. Lévai, Arkivoc, 2004, (vii), 15-33.
- S. Kamila, C. Mukherjee, T. K. Pradhan, A. De, Arkivoc, 2006, (ii), 45-60.
- M. T. Konieczny, W. Konieczny, S. Wolniewicz, K. Wierzba, Y. Suda, P. Sowinski, Tetrahedron, 2005, 61, 8648-8655.
- M. T. Konieczny, W. Konieczny, S. Okabe, H. Tsujimoto, Y. Suda, K. Wierzba, Chem. Pharm. Bull., 2006, 54, 350-353.
- M. T. Konieczny, W. Konieczny, Heterocycles, 2005, 65, 451-464.
- S. S. Liam, S. S. Reamonn, W. I. O’Sullivan, J. Chem. Soc., Perkin Trans., 1980, 1194-1198.
- W. Adam, D. Golsch, L. Hadjiarapoglou, A. Lévai, C. Nemes, T. Patonay, Tetrahedron, 1994, 50, 13113-13120.
- A. Lévai, Arkivoc, 2003, (xiv), 14-30.
- A. Lévai, T. Patonay, J. Heterocycl. Chem., 1999, 36, 747-753.
- Z. Dinya, I. Komaromi, F. Sztaricskai, A. Lévai, G. Litkei, Croat. Chem. Acta., 1993,
, 255-263.
- A. Boughaleb, G. Alhouari, B. Bennani, M. Daoudi, B. Garrigues, A. Kerbal, M. El yazidi, J. Soc. Chim. de Tunisie, 2010, 12, 109-115.
- A. Boughaleb, M. Akhazzane, G. Alhouari, B. Bennani, M. Daoudi, B. Garrigues, A. Kerbal, M. El yazidi, J. Soc. Chim. de Tunisie, 2011, 13, 117-122.
- A. Boughaleb, H. Zouihri, S. Gmouh, A. Kerbal, M. El yazidi, Acta Cryst., 2011, E67,
o1850-1850.
- A. Rössing, Ber., 1884, 17, 2988-3010.
- P. Perlmutter, Conjugated Addition Reaction in Organic Synthesis, Pergamon Press: Oxford, 1992, p.114.
- Z. Czarnocki, A. Siwicka, J. Szawkało, Curr. Org. Synth., 2005, 2, 301-331.
- B. E. Rossiter, N. M. Swingle, Chem. Rev., 1992, 92, 771-806.
- M. Bakhouch, G. Al Houari, M. El Yazidi, M. Saadi, L. El Ammari, Acta Cryst., 2014. E70, o587-587.
DOI: http://dx.doi.org/10.13171/mjc.4.1.2015.16.02.09.20/elyazidi
Refbacks
- There are currently no refbacks.
Copyright (c) 2015 Mediterranean Journal of Chemistry
