Bi- and polynuclear coordination compounds of d-elements as catalysts of homogeneous selective oxidation of thiol groups
Abstract
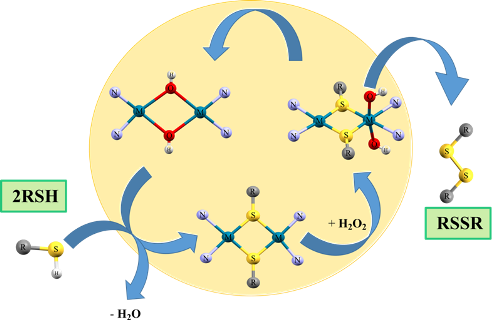
This research aims to study the catalytic cycle reactions for homogeneous selective oxidation of thiol RSH groups to RSSR with the participation of coordination compounds for d-element ions-NiII, PdII, Pt II, CuI , CuII. We used the DFT M06, PBE0 / Def2-TZVP methods to build the quantum chemical models of the reactions. We have developed a mechanism for the functioning of the catalytic system in which primary active centers are either binuclear {M(-OH)2M}n+ or polynuclear {M(μ-OH)2M(μ-OH)2M}2+ sites. Catalysts under consideration should retain stable spatial complementarity at all stages of the process. The main interrelated functions of the binuclear catalysts are the spatial approaching of anions RS– in the inner sphere of the bridged coordination compound required for the disulfide (–S–S–) cross-linking and the two-electron redox transfer during the transformation of these anions into disulfide (СH3)2S2
Full Text:
PDFReferences
M. Schieber, N.S. Chandel, ROS Function in Redox Signaling and Oxidative Stress, Curr. Biol, 2014, 24(10), 453-462.
S. J. Lippard, M. J. Berg, Principles of Bioinorganic Chemistry, University Science. Books, Mill Valley, California. 1994, 411
A. J. Montero, C. M. Diaz-Montero, Y. E. Deutsch, J. Hurley, L. G. Koniaris, T. Rumboldt, S. Yasir, M. Jorda, E. Garret-Mayer, E. Avisar, J. Slingerland, O. Silva, C. Welsh, K. Schuhwerk, P. Seo, M. D. Pegram, S. Gluck Phase 2 study of neoadjuvant treatment with NOV-002 in combination with doxorubicin and cyclophosphamide followed by docetaxel in patients with HER-2 negative clinical stage II-IIIc breast cancer, Breast Cancer Res. Treat., 2012, 132, 215-223
Patent RU 2417999 (2011). Balazovskii M.B. , Antonov V.G. , Belyaev A.N., Eremin A.V.
D. M. Easton, A. Nijnik, M. L. Mayer, R. E. Hancock, Potential of immunomodulatory host defense peptides as novel anti-infectives, Trends in Biotech., 2009, 27(10), 582-590.
D. M. Townsend, L. He, S. Hutchens, T. E. Garrett, C. J. Pazoles, K. D. Tew, NOV-002, a glutathione disulfide mimetic, as a modulator of cellular redox balance, Canc. Res., 2008, 68(8), 2870-2877.
J. D. Uys, Y. Manevich, L. C. DeVane, L. He, T. E. Garret, C. J. Pazoles, D. M. Townsend, Preclinical pharmacokinetic analysis of NOV-002, a glutathione disulfide mimetic, Biomed. & Pharmacother., 2010, 64(7), 493-498.
Patent RU 2451010 (2012). Balazovskii M.B., Antonov V.G., Belyaev A.N., Eremin A.V.
W. P. Jencks, Catalysis in Chemistry and Enzymology, Courier Corporation, 1987.
J. A. Campbell, General Chemistry, 1949.
J. H. Espenson, Chemical Kinetics and Reaction Mechanisms 2nd ed., McGraw-Hill, 2002, 156-160.
Y. Zhao, D. G. Truhlar, Theor. Chem. Account, 2008, 120, 215, CrossRef| CAS| Web of Scienceé Times Cited, 168.
J. P. Perdew, K. Burke, M. Ernzerhof, Generalized gradient approximation made simple, Phys. rev. let., 1996, 77(18), 3865.
P. J. Hay, T. H. Dunning Jr, Modern Theoretical Chemistry, Vol. 3. Methods of Electronic Structure Theory. Schaefer III, HF (Ed.) Plenum, New York, 1976, 1.
M. Dolg, U. Wedig, H. Stoll, H. Preuss, Energy-adjusted abinitio pseudopotentials for the first row transition elements. The J. of Chem. Phys., 1987, 86(2), 866-872.
D. Andrae, U. Haeussermann, M. Dolg, H. Stoll, H. Preuss, Energy-adjustedab initio pseudopotentials for the second and third row transition elements. Theor. Chem. Accounts: Theory, Computation, and Modeling (Theoretica Chimica Acta), 1990, 77(2), 123-141.
A. Frisch, M. J. Frisch, F. R. Clemente, G. W. Trucks, Gaussian 09 User's Reference, Version 8.0. Gaussian: Wallingford, CT, 2009.
F. Weigend, R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. And Chem. Phys., 2005, 7(18), 3297-3305.
G. A. Zhurko, D. A. Zhurko, ChemCraft, version 1.6.
URL: http://www.chemcraftprog.com, 2009.
S. Miertuš, E. Scrocco, J. Tomasi, Electrostatic interaction of a solute with a continuum. A direct utilizaion of AB initio molecular potentials for the prevision of solvent effects, Chem. Phys., 1981, 55(1), 117-129.
J. Tomasi, M. Persico, Molecular interactions in solution: an overview of methods based on continuous distributions of the solvent, Chem. Rev., 1994, 94(7), 2027-2094.
R. G. Pearson, Hard and soft acids and bases, ACS, 1963, 85(22), 3533-3539.
F. O. Rice, E. Teller, The role of free radicals in elementary organic reactions, J. Chem. Phys., 1938, 6(8), 489-496.
B. Lippert, C. J. L. Lock, B. Rosenberg, M. Zvagulis, Hydroxo-bridged platinum (II) complexes. 4. Crystal structure and vibrational spectra of di-μ-hydroxo-bis [diammineplatinum(II)] carbonate dihydrate, [(NH3)2Pt(OH)2Pt(NH3)2](CO3)•2H2O, Inorg. Chem., 1978, 17(11), 2971-2975.
L. Pauling, Nature of forces between large molecules of biological interest, Nature, 1948, 161(4097), 707-709.
D. E. Metzler, Biochemistry: the chemical reactions of living cells. Academic Press, 2003.
M. L. Tobe, Reaction mechanisms in inorganic chemistry, (Inorganic chemistry, series two), University Park Press, 1974, 380.
S. Ahmad, A. Espinosa, T. Ahmad, M. Sohail, A. A. Isab, M. Saleem, É. de las Heras, Synthesis, theoretical calculations and antimicrobial studies of copper (I) complexes of cysteamine, cysteine and 2-mercaptonicotinic acid, Polyhedron, 2015, 85, 239-245.
A. Farazdel, M. Dupuis, On the determination of the minimum on the crossing seam of two potential energy surfaces, J. Comp. Chem., 1991, 12(2), 276-282.
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, S. T. Elbert, M. S. Gordon, J. H. Jensen, T. L. Windus, Iowa State University, J. Comp. Chem, 1993, 14, 1347.
R. J. Gillespie, I. Hargittai, The VSEPR Model of Molecular Geometry Allyn and Bacon, Newton, MA, 1991.
DOI: http://dx.doi.org/10.13171/mjc73181017-eremin
Refbacks
- There are currently no refbacks.
Copyright (c) 2018 Mediterranean Journal of Chemistry
