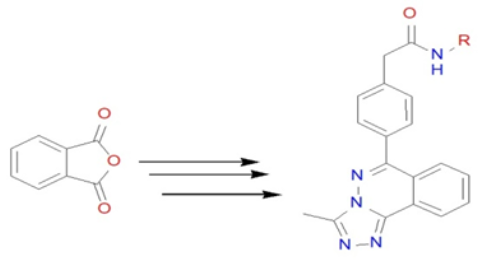
Synthesis of new analogs of 3-methyl-[1,2,4] triazolo [3,4-a] phthalazines via Suzuki coupling and evaluation of their anticancer and antimicrobial activity
Abstract
A series of new N-aryl substituted phenyl acetamide analogs of 3-methyl-[1,2,4] triazolo[3,4-a] phthalazines were synthesized starting from commercially available, in-expensive phthalic anhydride in good yields (65-75 %) via Suzuki Coupling. These compounds were tested for inhibition activity against HCT 116 cancer cell line by using MIT assay. Among the library of compounds, N-(3-methoxyphenyl)-2-(4-(3-methyl-[1,2,4]triazolo[3,4-a]phthalazin-6-yl)phenyl) acetamide followed by 2-(4-(3-methyl-[1,2,4]triazolo[3,4-a]phthalazin-6-yl)phenyl)-N-(m-tolyl) acetamide and  N-(3-chlorophenyl)-2-(4-(3-methyl-[1,2,4]triazolo[3,4-a]phthalazin-6-yl)phenyl) acetamide  were found to be active compounds with IC50 of 70 to and 90 µg mL-1. Further, the compounds were also screened for their antimicrobial activities.
Full Text:
PDFReferences
- R. Siegel, C. De Santis, A. Jemal, Colorectal cancer statistics – 2014, CA Cancer J., 2014, 64(2), 104.
- World Cancer Report (2014).
- D. E. Chavez, D. A. Parrish New heterocycles from tetrazines and oxadiazoles, J. Heterocycl. Chem., 2009, 46, 88-90.
- G. Bold, K. H. Altmann, J. Frei, M. Lang, P. W. Manley, P. Traxler, B. Wietfeld, J. Bruggen, E. Buchdunger, R. Cozens, S. Ferrari, P. Furet, F. Hofmann, G. Martiny-Baron, J. Mestan, J. Rosel, M. Sills, D. E. L. Stover, M. A. A. Hashash, F. Acemoglu, E. Boss, R. Emmenegger, L. Lasser, E. Masso, R. Roth, C. Schlachter, W. Vetterli, D. Wyss, J. Wood, M, New anilinophthalazines as potent and orally well-absorbed inhibitors of the VEGF receptor tyrosine kinases useful as antagonists of tumour-driven angiogenesis, J. Med. Chem., 2000, 43, 2310-2323.
- G. Strappaghetti, C. Brodi, G. Giannaccini, L. Betti, New 4-(4-methyl-phenyl) phthalazine-1(2H)-one derivatives and their effects on α1-receptors, Bioorg. Med. Chem. Lett., 2006, 16, 2575-2579.
- A. D. Lebsack, J. Gunzner, B. Wang, R. Pracitto, H. Schaffhauser, A. Santini, J. Aiyar, R. Bezverkov, B. Munoz, W. Liuc, S. Venkatraman, Identiï¬cation and synthesis of [1,2,4]triazolo[3,4-a]phthalazinederivatives as high-affinity ligands to the α2δ-1 subunit of voltage-gated calcium channel, Bioorg. Med. Chem. Lett., 2004, 14, 2463-2467.
- T. Haack, R. Fattori, M. Napoletano, F. Pellacini, G. Fronza, G. Raffaini, F. Ganazzoli, Phthalazine PDE IV inhibitors: a conformational study of some 6-methoxy-1,4-disubstituted derivatives, Bioorg. Med. Chem., 2005, 13, 4425-4433
- E. L. Piatnitski, M. A. J. Duncton, A. S. Kiselyov, R. Katoch-Rouse, D. Sherman, D. L. Milligan, C. Balagtas, W. C. Wong, J. Kawakami, J. F. Doody, Arylphthalazines: Identification of a new phthalazine chemotype as inhibitors of VEGFR kinase, Bioorg. Med. Chem. Lett., 2005, 15, 4696-4698.
- K. A. Menear, C. Adcock, F. C. Alonso, K. Blackburn, L. Copsey, J. Drzewiecki, A. Fundo, A. Le Gall, S. Gomez, H. Javaid, C. F. Lence, N. M. B. Martin, C. Mydlowski, G. C. M. Smith, Novel alkoxybenzamide inhibitors of poly (ADP-ribose) polymerase, Bioorg. Med. Chem. Lett., 2008, 18, 3942-3945.
- C. K. Ryu, E. R. Park, Mi-Young Ma, Ji-HeeNho, Synthesis and antifungal activity of 6-arylamino-phthalazine-5,8-diones and 6,7-bis(arylthio)-phthalazine-5,8-diones, Bioorg. Med. Chem. Lett., 2007, 17, 2577-2580.
- C. A. Gandolï¬, G. Beggiolin, E. Menta, M. Palumbo, C. Sissi, S. Spinelli, F. Johnson, Chromophore-Modified Antitumor Anthracenediones: Synthesis, DNA Binding, and Cytotoxic Activity of 1,4-Bis[(aminoalkyl)amino] benzo[g]phthalazine-5,10-diones, J. Med. Chem., 1995, 38, 526-536.
- E. M. Kassem, M. M. Kamel, M. El-zahar, New 1(2H)-phthalazinones of potential antimicrobial activity, Pharmazie, 1990, 45, 215-216.
- St. Dima, M. Caprosu, M. Ungureanu, G. Grosu, M. Petrovanu, Nouveaux derives de la 1a 1-methyl-phthalazinium ylides doués d’activités antimicrobiennes et fongistatiques, Ann. Pharm. Fr., 1999, 57, 415-416.
- S. Ito, K. Yamaguchi, Y. Komoda, Structural Confirmation of the Nitration Product of the 1(2H)-Phthalazinone as the 2-Nitro-1(2H)-phthalazinone, Chem. Pharm. Bull., 1992, 40, 3327-3329.
- T. Haack, R. Fattori, M. Napoletano, F. Pellacini, G. Fronza, G. Raffaini, F. Ganazzoli, Phthalazine PDE IV inhibitors: Conformational study of some 6-methoxy-1,4-disubstituted derivatives, Bioorg. Med. Chem., 2005, 13, 4425-4433.
- G Heinisch, and H. Frank, Pharmacologically Active Pyridazine Derivatives part 1: Progress in Medicinal Chemistry; ed. By G.P. Ellis; Elsevier: Amsterdam, The Netherlands 1990, 27, pp. 1-50.
- G Heinisch, and H. Frank, Pharmacologically Active Pyridazine Derivatives part 2: Progress in Medicinal Chemistry; ed. By G.P. Ellis; Elsevier: Amsterdam, The Netherlands 1992, 29, pp. 141-184.
- A. Melikian, G. Schiewer, J. P. Chambon, C. G. Wermuth, Condensation of muscimol or thiomuscimol with aminopyridazines yields GABA-A antagonists, J. Med. Chem., 1992, 35, 4092-4097.
- M. Napoletano, G. Norcini, F. Pellacini, G. Morazzoni, P. Ferlenga, L. Pradella, Phthalazine PDE4 inhibitors. Part 2: The synthesis and biological evaluation of 6-methoxy-1,4-disubstituted derivatives, Bioorg. Med. Chem. Lett., 2001, 11, 33-37.
- Jill. K. Robinson, Mark. J. Bollinger, and W. M. John, Luminol/H2O2 Chemiluminescence Detector for the Analysis of Nitric Oxide in Exhaled Breath, Anal. Chem., 1999, 22, 5131-5136.
- H. Arakawa, J. IshidaJ, M. Yamaguchi, M. Nakamura, Chemiluminescent Products of Reaction between α-Keto Acids and 4, 5-Diaminophthalhydrazide, Chem. Pharm. Bull. , 1990, 38, 3491-3493.
- H. Arakawa, J. IshidaJ, M.Yamaguchi, M. Nakamura, A New Chemiluminogenic Substrate for N-Acetyl-β-D-glucosaminidase, 4'-(6'-Diethylaminobenzofuranyl)phthalylhydrazido-N-acetyl-β-D-glucosaminide, Chem. Pharm. Bull. , 1991, 39: 411-416.
- K. Samoto, Y. Ohkura, Ring expansion reaction of 1,3-dithiolanes and 1,3-oxathiolanes using tellurium tetrachloride, Chem. Lett., 1990, 19, 1323-1326.
- R. T. Buckler, H. E. Hartzler, E. Kurchacova, G. Nichols, B. M. Phillips, Synthesis and antiinflammatory activity of some 1,2,3- and 1,2,4-triazolepropionic acids, J. Med. Chem., 1978, 21, 1254-1260.
- R. Alvarez, S. Velazquez, A. S. Felix, S. Aquaro, E. D. Clercq, C. F. Perno, A. Karlsson, J. Balzarini, M. J. Camarasa, 1,2,3-Triazole-[2,5-Bis-O-(tert-butyldimethylsilyl)-Beta-D-ribofuranosyl]-3'-spiro-5''-(4''-amino- 1'',2''-oxathiole 2'',2''-dioxide) (TSAO) Analogs: Synthesis and Anti-HIV-1 Activity, J. Med. Chem., 1994, 37, 4185-4195.
- M. J. Genin, D. A. Allwine, D. J. Anderson, M. R. Barbachyn, D. Edward Emmert, S. A. Garmon, D. A. Graber, K. C. Grega, J. B. Hester, D. K. Hutchinson, J. Morris, R. J. Reischer, C. W. Ford, G. E. Zurenko, J. C. Hamel, R. D. Schaad, D. Stapert, B. H. Yagi, Substituent Effects on the Antibacterial Activity of Nitrogen−Carbon-Linked (Azolylphenyl)oxazolidinones with Expanded Activity Against the Fastidious Gram-Negative Organisms Haemophilus influenza and Moraxella catarrhalis, J. Med. Chem., 2000, 43, 953-970.
- F. Reck, F. Zhou, M. Girardot, G. Kern, C. J. Eyermann, R. R. Ramsay, N. J. Hales, M. B. Gravestock, Identification of 4-Substituted 1,2,3-Triazoles as Novel Oxazolidinone Antibacterial Agents with Reduced Activity against Monoamine Oxidase A, J. Med. Chem., 2005, 48, 499-506.
- A. C. Cunha, J. M. Figueiredo, J. L. M. Tributino, A. L.P. Miranda, H. C. Castro, R. B. Zingali, C. M. A. Fraga, M. C. B. V. de Souza, V. F. Ferreira, E. Barreiro, Antiplatelet properties of novel N-substituted-phenyl-1,2,3-triazole-4-acyl hydrazone derivatives, J. Bioorg. Med. Chem., 2003, 11, 2051-2059.
- S. A. Shahzad, Md. Yar, Z. A. Khan, L. Shahzadi, Sd. Ali. R. Naqvi, A. Mahmood, S. Ullah, A. J. Shaikh, T. A. Sherazi, A. T. Bale, J. Kukułowicz, M. Bajda, Identification of 1,2,4-triazoles as new thymidine phosphorylase inhibitors: Future anti-tumour drugs, bioorganic chemistry, 2019, 85, 209-220.
- S. Akin, H. Ayaloglu, E. Gultekin, A. Colak, O. Bekircan, M. Y. Akatin, Synthesis of 1,2,4-triazole-5-on derivatives and determination of carbonic anhydrase II isoenzyme inhibition effects, bioorganic chemistry, 2018, 83, 170-179.
- J. Chen, Z. Ding, Y. Peng, F. Pan, Li. Jianjun, L. Zou, Y. Zhang, H. Liang, HIF-1α Inhibition Reverses Multidrug Resistance in Colon Cancer Cells via Down regulation of MDR1/P-Glycoprotein, Plos. One., 2014, 9, e98882.
- K. Harris, X. Gao, S. Huerta-Yepez, P. Kapur, S. Huerta, HIF-1α and Resistance to Ionizing Radiation in Rectal Cancer, Surgery Curr. Res., 2014, 4, 200.
- N. Ramesh, M. Gangadhara Rao, V. Ravi, V. Umamaheswara Rao, B. Hari Babu, Mercuric chloride catalyzed synthesis of some anticancer 2-aryl-2,3-dihydroquinizolin-4(1H)-ones, Med Chem Res., 2016, 25, 1945-1951.
- N. Ramesh, M. Gangadhara Rao, M. Tirumala, V. Umamaheswara Rao, B. Hari Babu, Synthesis, Anticancer and Antioxidant Evaluation of Some New 2-Aryl and 2-Pyrazole-2,3-dihydroquinazolin-4(1H)-ones, Asian J. Chem., 2016, 28, 1321-1324.
- O. Surendranatha Reddy, Ch. Venkata Surya Narayana, K. J. P. Narayana, V. Anuradha, B. Hari Babu, Synthesis and cytotoxic evaluation for some new 2,5-disubstituted pyrimidine derivatives for anticancer activity, Med. Chem. Res., 2015, 24, 1777-1788.
- O. Surendranatha Reddy, Ch. Venkata Surya Narayana, N. Sharmila, G. V. Ramana, V. Anuradha, B. Hari Babu, Synthesis and Cytotoxic Evaluation for Some New Dihydropyrimidinone Derivatives for Anticancer Activity, Lett. Drug Des. Discov., 2013, 10, 699-705.
- Q. R. Zhang, D. Q. Xue, P. He, K. P. Shao, P. J. Chen, Y. F. Gu, J. L. Ren, L. H. Shan, H. M. Liu, Synthesis and antimicrobial activities of novel 1,2,4-triazolo [3,4-a] phthalazine derivatives, Bioorg. & Med. Chem. Lett., 2014, 24, 1236-1238.
- X. Y. Sun, C. Hu, X. Q. Deng, C. X. Wei, Z. G. Sun, Z. S. Quan, Synthesis and anti-inflammatory activity evaluation of some novel 6-alkoxy(phenoxy)-[1,2,4] triazolo[3,4-a]phthalazine-3-amine derivatives, Eur. J. Med. Chem., 2010, 45, 4807-4812.
- D. Q. Xue, X. Y. Zhang, C. J. Wang, L. Y. Ma, N. Zhu, P. He, K. P. Shao, P. J. Chen, Y. F. Gu, X. S. Zhang, C. F. Wang, C. H. Ji, Q. R. Zhang, H. M. Liu, Synthesis and anticancer activities of novel 1,2,4-triazolo[3,4-a] phthalazine derivatives, Eur. J. Med. Chem., 2014, 85, 235- 244.
- J G Cappucino, N. Sherman, Microbiology, a laboratory manual; ed. by K. Churchman; Pearson Education Inc; New Delhi, 2004, pp. 282.
DOI: http://dx.doi.org/10.13171/mjc841905257hbb
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
