Investigation of pipes and sprinklers scaling at the golf course turf irrigated by treated wastewater of Mzar plant in Agadir-Morocco
Abstract
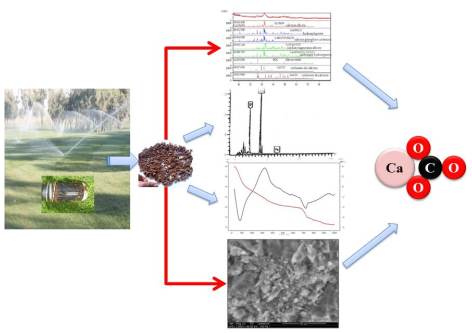
In Agadir city, the reuse of treated wastewater for irrigation of green spaces has faced the problem of scaling of the pipes of these waters. This research paper aims at studying the phenomenon of scaling caused by the treated wastewater from the Mzar sewage treatment plant in the city of Agadir in south-west of Morocco. These waters are used in the irrigation of golf turf “Ocean”. The formation of solid deposits in the irrigation systems has decreased their lifetime, and consequently, a loss of load and performance is occurring. Thus, the sprinklers used in golf turf irrigation are plugged in the first weeks of the beginning of their operation. The wastewater used for the irrigation of the golf was analyzed at various points using physicochemical measurements: in the golf entry, at the tarpaulin and the exit of the sprinklers. The samples of treated wastewater and obtained scale were studied. The characterization of the scale samples formed in the passageways of the treated wastewaters has been performed by X-ray fluorescence spectrometry, X-ray diffraction, thermogravimetric analysis, differential thermal analysis and scanning electron microscopy. The results of the physicochemical analysis of the waters show that they are full of bicarbonates (653 mg/L), chloride (478 mg/L), nitrate (412 mg/L), sodium (425 mg/L) and in calcium (199 mg/L). Their pH is slightly alkaline. The analysis of the scale reveals that it is rich in calcium and phosphorus. It is formed of calcium carbonate (CaCO3), silica (SiO2), calcium silicate (Ca2SiO4), hydroxylapatite (Ca10P6O26H2), calcium carbonate-apatite(Ca10 (PO4) 6CO3) and silicate calcium and magnesium (Ca5MgSi3O12).
Full Text:
PDFReferences
- E. Ibariouen, La réutilisation des eaux usées épurées dans le Grand-Agadir, Régie Autonome Multi-Services d’Agadir, Les cahiers d’échange Med N°8. XIVème Rencontre Internationale d’Echanges Méditerranéens au Maroc. 25/11 au 02/12/2015.
- B. Boudinar, H. Mouhanni, M. Houari, A. Bendou, Analyses physico-chimiques du gazon irrigué par les eaux usées épurées de la station Mzar du grand Agadir- Maroc, revue internationale d'héliotechnique, 2015, 46, 1-7.
- Programme mixte FAO/UNW-DPC/UNU-INWEH. Montage Institutionnel et analyse environnementale et socioéconomique et concernant le projet de réutilisation des eaux usées épurées. Rapport global de synthèse. Mars 2012.
- Direction des affaires intérieures, Compétitivité économique du Maroc ébauche de révision des normes de qualité des eaux usées traitées destinées à l’irrigation des cultures et a l’arrosage des espaces verts USAID. Mars 2013.
- USAID. Réutilisation des Eaux Usées en Irrigation. United States Agency for International Development.projet de « Pérennité des Ressources en Eau du Maroc –PREM ».
- A. Kerfati, Réutilisation en irrigation des eaux usées au Maroc, DIAEA / Ministère de l’Agriculture. Agadir. 7-11 décembre 2009.
- A. Hadfi, S. Ben Aazza, M. Belattar, S. Mohareb, A. Driouiche, “Evaluation of the irrigation water quality in Biougra circle along with highlighting the effectiveness of a scaling inhibitor”, Mediterranean Journal of Chemistry, 2018.7(4), 272-285.
-A. Kamari, F. Gharagheizi, A.Bahadori, A.H. Mohammadi, Determination of the Equilibrated Calcium Carbonate (Calcite) Scaling in Aqueous Phase Using a Reliable Approach, Journal of the Taiwan Institute of Chemical Engineers, 2014, 45, 1307-1313.
-A. Korchef, M. Touaibi, Effect of pH and temperature on calcium carbonate precipitation by CO2 removal from iron-rich water, Water and Environment Journal. 2019. https://doi.org/10.1111/wej.12467.
-R. Mimouni, B. Yacoubi, R. Eddabra, Physicochemical quality of wastewater purified by infiltration-percolation: case of the plant of Ben Sergao (south-western Morocco) after ten years of running, Rev. Microbiol. Ind. San et Environn., 2011, 5(1), 101-114.
-J. Yin, X. Kang, C. Qin, B. Feng, A. Veeraragavan, D. Saulov, Modeling of CaCO3 Decomposition under CO2/H2O Atmosphere in Calcium Looping Processes, Fuel Processing Technology, 2014, 125, 125-138.
-N. Hafid, M. Belaatar, S. Ben-Aazza, A. Hadfi, M. Ezahri, A. Driouiche, Characterization of Scale Formed in Drinking Water and Hot Water Pipes in the Taliouine Downtown-Morocco, American Journal of Analytical Chemistry, 2015, 6, 677-686.
-M.N. Freire, J.N.F. Holanda, Characterization of avian eggshell waste aiming its use in a ceramic wall tile paste, Cerâmica, 2006, 52(324),
-244.
-S. Serena, M.A. Sainz, A. Caballero, Single-phase silico-carnotite synthesis in the subsystem Ca3(PO4)2-Ca2SiO4, Ceramics International, 2014, 40, 8245-8252.
-N. Yamaguchi, Y. Masuda, Y. Yamada, H. Narusawa, C. Han-Cheol, Y. Tamaki, T. Miyazaki, Synthesis of CaO-SiO2 Compounds Using Materials Extracted from Industrial Wastes, Open Journal of Inorganic Non-Metallic Materials, 2015, 5, 1-10.
-C. Remy, B. Reynard, M. Madon, Raman spectroscopic investigations of dicalcium silicate: Polymorphs and high-temperature phase transformations, Journal of the American Ceramic Society, 1997, 80(2), 413-423.
-Z. Gou, J. Chang, Synthesis and in vitro bioactivity of dicalcium silicate powders, Journal of the European Ceramic Society, 2004, 24, 93-99.
DOI: http://dx.doi.org/10.13171/mjc960200104984ah
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
