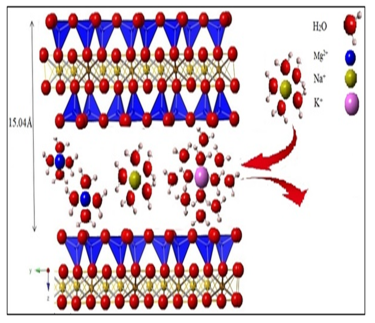
Calcite, the main corrosion inhibitor contained in the raw clay (Rhassoul) of brass in 3% NaCl medium.
Abstract
Full Text:
PDFReferences
- M.H. Hussin, M.J. Kassim, The corrosion inhibition and adsorption behavior of Uncaria gambir extract on mild steel in 1 M HCl, Mater. Chem. Phys, 2011, 125, 461-468.
- F. Mansfeld, T. Smith, E. P. Parry, Benzotriazole as corrosion inhibitor for copper, Corrosion, 1971, 27, 289- 294.
- E. Geler, D. S. Azambuja, Corrosion inhibition of copper in chloride solutions by pyrazole, Corros. Sci, 2000, 42, 631-643.
- R. Ravichandran, N. Rajendran, Influence of benzotriazole derivatives on the dezincification of 65-35 brass in sodium chloride, Applied Surface Science, 2005, 239, 182-192.
- Z. Mountassir, A. Srihari, Electrochemical behaviour of Cu-40Zn in 3% NaCl solution polluted by sulphides: effect of aminotriazole, Corros. Sci, 2007, 49, 1350-1361.
- T. Kosec, D. K. Merl, I. Milošev, Impedance and XPS study of benzotriazole films formed on copper, copper-zinc alloys and zinc in chloride solution, Corros. Sci, 2008, 50, 1987-1997.
- J. R. Xavier, S. Nanjundan, N. Rajendran, Electrochemical adsorption properties and inhibition of brass corrosion in natural seawater by thiadiazole derivatives: experimental and theoretical investigation, Industrial & Engineering Chemistry Research, 2012, 51, 30-43.
- S. Langard, T. Vigander, Occurrence of lung cancer in workers producing chromium pigments, Occupational and Environmental Medicine, 1983, 40, 71-74.
- S. A. Katz, H. Salem, The toxicology of chromium with respect to its chemical speciation, J Appl Toxicol, 1993, 13, 217-224.
- S. Tazi, I. Raissouni, F. Chaouket, D. Bouchta, A. Dahdouh, R. Elkhamlichi, H. Douhri, The Inhibition effect of Brass corrosion in NaCl 3% by Eugenol, J. Mater. Environ. Sci, 2016, 7, 1642-1652, https://www.jmaterenvironsci.com/ Document/ vol 7/vol 7_N5/181-JMES-2120-Tazi.pdf.
- A. Chraka, I. Raissouni, N. Ben Seddik, S. Khayara, A. Ibn Mansour, H. Belcadib, F. Chaouket, D. Bouchta, Aging time effect of Ammi visnaga (L.) lam essential oil on the chemical composition and corrosion inhibition of brass in 3% NaCl medium. Experimental and theoretical studies, Materials Today: Proceedings, https://doi.org/10.1016/j. matpr.2019.08.086
- M. Y. Lvov, G. D. Shchukin, H. Mohwald, R. R. Price, Halloysite Clay Nanotubes for Controlled Release of Protective Agents, J. Am. Chem. Soc, 2008, 2, 814-820.
- E. Abdullayev, Y. M. Lvov, Halloysite clay nanotubes for controlled release of protective agents, Journal of Nanoscience and Nanotechnology, 2011, 11, 10007-10026.
- E. Shchukina, D. Shchukin, D. Grigoriev, Effect of inhibitor-loaded halloysites and mesoporous silica nanocontainers on corrosion protection of powder coatings, Progress in Organic Coatings, 2017, 102, 60-65.
- A. Ait Aghzzaf, B. Rhouta, J. Steinmetz,
E. Rocca, L. Aranda, A. Khalil, J. Yvon. L. Daoudi, Corrosion inhibitors based on chitosan-heptanoate modified beidellite, Applied Clay Science, 2012, 65-66, 173-178.
- A. Ait Aghzzaf, B. Rhouta, E. Rocca, A. Khalil, C. Caillet, R. Hakkou, Heptanoic acid adsorption on grafted palygorskite and its application as controlled-release corrosion inhibitor of steel, Materials Chemistry and Physics, 2014, 148, 335-342.
- B.L. Yu, X. L. Pan, J. Y. Uan, Enhancement of corrosion resistance of Mg-9 wt.% Al-1 wt.% Zn alloy by a calcite (CaCO3) conversion hard coating, Corros. Sci, 2010, 52, 1874-1878.
- A. Ait Aghzzaf, B. Rhouta, E. Rocca, A. Khalil, Grafted palygorskite as containers of heptanoate for corrosion protection of steel in NaCl medium, Corros. Sci, 2017, 114, 88-95.
- L. Bouna, B. Rhouta. M. Amjoud, A. Jada, F. Maury, L. Daoudi, F. Senoq, Correlation between electrokinetic mobility and ionic dyes adsorption of Moroccan stevensite, Applied Clay Science, 2010, 48, 527-530.
- A. Benhammou, B. Tanouti, L. Nibou, A. Yaacoubi, J. P. Bonnet, Mineralogical and physicochemical investigation of Mg-smectite from Jbel Ghassoul, Morocco, Clays and Clay Minerals, 2009, 57, 264-270.
- S. Caillère, S. Henin, M. Rautureau, Minéralogie des Argiles : Classification et Nomenclature, Ed., Masson et INRA, Paris, 1982.
- B. Rhouta, H. Kaddami, J. Elbarqy, M. Amjoud, L. Daoudi, F. Maury, F. Senocq, A. Maazouz, J. F. Gerard, Elucidating the crystal-chemistry of Jbel Rhassoul stevensite (Morocco) by advanced analytical techniques, Clay minerals, 2008, 43, 393-404.
- T. Holtzapffel, Les mineraux argileux: préparation, analyse diffractométrique et détermination, Ed., Société Géologique du Nord, 1985.
- A. Benhammou, A. Yaacoubi, L. Nibou, B. Tanouti, Adsorption of metal ions onto Moroccan stevensite: kinetic and isotherm studies, Journal of Colloid and Interface Science, 2005, 282, 320-326.
- L. Bouna, B. Rhouta, M. Amjoud, F. Maury, M. Jada, L. Daoudi, F. Senocq, M. C. Lafont, C. Drouet, Synthèse, caractérisations et tests photocatalytiques d’un matériau argileux d’origine naturelle à base de beidellite fonctionnalisée par TiO2, Matériaux & Techniques, 2012, 100, 241-252,
- N. Ben Seddik, I. Raissouni, K. Draoui, A. Ait Aghzzaf, A. Chraka, B. Aznag, F. Chaouket, D. Bouchta, Anticorrosive performance of lanthanum ions intercalated Stevensite clay on brass in 3% NaCl medium,
Materials Today: Proceedings, https://doi.org/10.1016/j.matpr.2019.08.085
- T. Theivasanthi, M. Alagar, physics.chem-ph, Titanium dioxide (tio2) nanoparticles xrd analyses: An insight, 2013, arXiv:1307.1091.
- S. Chanel, N. Pébère, An investigation on the corrosion of brass-coated steel cords for tyres by electrochemical techniques, Corros. Sci. 2001, 43, 413-427.
- A. Ait Aghzzaf, B. Rhouta, E. Rocca, A. Khalil, J. Steinmetz, Corrosion inhibition of zinc by calcium exchanged beidellite clay mineral: A new smart corrosion inhibitor, Corros. Sci, 2014, 80, 46-52.
- E. S. Ferreira, F. C. Giacomelli, A. Spinelli, Evaluation of the inhibitory effect of L-ascorbic acid on the corrosion of mild steel, Mater. Chem. Phys, 2004, 83, 129-134.
- I. Aiad, M. M. El-Sukkary, E. A. Soliman, M. Y. El-Awady, S. M. Shaban, Inhibition of mild steel corrosion in acidic medium by some cationic surfactants, J. Ind. Eng. Chem, 2014, 20, 3524-3535.
- C. Des louis, B. Tribollet, G. Mengoli, M. Musiani, Electrochemical behaviour of copper in neutral aerated chloride solution. I. Steady-state investigation, J. Applied Electrochem, 1988, 18, 374-383.
- Z. Avramovic, M. Antonijevic, Corrosion of cold-deformed brass in acid sulphate solution, Corros. Sci, 2004, 46, 2793-2802.
- I. B. Obot, N. O. Obi-Egbedi, Anti-corrosive properties of xanthone on mild steel corrosion in sulphuric acid: Experimental and theoretical investigations, Current Applied Physics, 2011, 11, 382-392.
- M. Bitenc, M. Marinsek, Z. C Orel, Preparation and characterization of zinc hydroxide carbonate and porous zinc oxide particles., J. Eur. Ceram. Soc, 2008, 28, 2915-2921.
- M. R. Mahmoudian, W. J. Basirun, Y. Alias,
M. Ebadi, Synthesis and characterization of polypyrrole/Sn-doped TiO2 nanocomposites (NCs) as a protective pigment, Appl. Surf. Sci, 2011, 257, 8317-8325.
DOI: http://dx.doi.org/10.13171/mjc93191016530nbs
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
