A DFT reinvestigation of chemo- and stereoselectivity epoxidation from α- and ɣ-trans himachalene with meta Chloroperoxybenzoic acid
Abstract
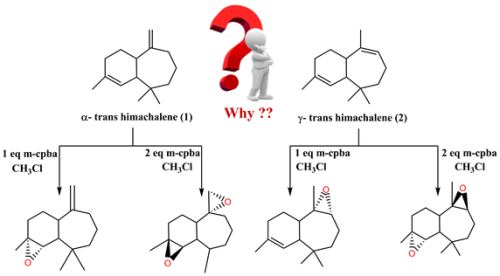
In this work the epoxidation reaction of the α- and ɣ-trans himachalene in the presence of meta chloroperoxybenzoic acid (m-CPBA) has been studied within the Density Functional Theory (DFT) method at the B3LYP/6-311G(d,p) level in dichloromethane as a solvent, in order to shed light on the chemo- and stereoselectivity in the course of the reaction. Analysis of the Conceptual Density Functional Theory (CDFT) reactivity indices indicate that the m-CPBA will behave as electrophilic while α- and ɣ-trans himachalene will behave as a nucleophile and the attacks observed experimentally are correctly predicted by the electrophilic Pk + and nucleophilic Pk - Parr functions. The two reactive paths associated with chemo and stereoselectivity approach modes of m-CPBA on C=C reactive sites in α and ɣ-trans himachalene have been analyzed. They showed that m-CPBA reacted as electrophile whereas α- and ɣ- trans himachalene as a nucleophile. The Monoepoxidation of α- and ɣ- trans himachalene leads to the formation of two stereoisomers, on the most substituted double bond "C=C», one of the two is a majority. The diepoxidation reaction of α- and ɣ- trans h
Full Text:
PDFReferences
- F. Couic-Marinier, A. Lobstein, Les huiles essentielles gagnent du terrain à l’officine, Actualités pharmaceutiques, 2013, 52, 18-21.
- A. Chekroun, A. Jarid, A. Benharref, A. Boutalib, Computational study of chemo-and stereoselectivity of α-cis, α-trans and α′-trans-himachalene epoxidation by MCPBA, Journal of Molecular Structure: THEOCHEM, 2002, 588, 201-210.
- M. Plattier, P. Teisseire, Essential oil of the Atlas cedar, Recherches, 1974, 19, 131-144.
- A. Chiaroni, C. Riche, A. Benharref, H. El Jamili, E. Lassaba, 2α, 3α: 7β, 13β-Diepoxy-trans-himachalane, Acta Crystallographica Section C: Crystal Structure Communications, 1995, 51, 1171-1173.
- A. Chiaroni, C. Riche, A. Benharref, H. El Jamili, E. Lassaba, 2α, 3α: 7β, 8β-Diépoxy-trans-himachalane, Acta Crystallographica Section C: Crystal Structure Communications, 1996, 52, 2502-2504.
- M. A. Loriot, P. Beaune, La vitamine K époxyde réductase: du sang neuf dans les traitements anticoagulants oraux, La Revue de médecine interne, 2006, 27, 979-982.
- M. R. Loizzo, A. M. Saab, G. A. Statti, F. Menichini, Composition and α-amylase inhibitory effect of essential oils from Cedrus libani, Fitoterapia, 2007, 78, 323-326.
- M. Aziz, F. Rouessac, Syntheses en serie racemique et en serie optiquement active d'une famille de derives oxygenes naturels de l'ombelliferone. Structure spatiale du (-) epoxy-3'6'auraptene, Tetrahedron, 1988, 44, 101-110.
- B. Frérot, M. Renou, C. Malosse, C. Descoins, Isolement et identification de composés à activité phéromonale chez les femelles de l'arctiide Tyria jacobaeae: détermination biologique de la configuration absolue du composé majoritaire, Entomologia experimentalis et applicata, 1988, 46, 281-289.
- B. Satrani, M. Aberchane, A. Farah, A. Chaouch, M. Talbi, Composition chimique et activité antimicrobienne des huiles essentielles extraites par hydrodistillation fractionnée du bois de Cedrus atlantica Manetti, Acta botanica gallica, 2006, 153, 97-104.
- G. Venkatesh, M. Govindaraju, P. Vennila, Experimental and theoretical spectral investigations of 5-chloro-ortho-methoxyaniline using FT-IR, FT-Raman and DFT analysis, Indian. J. Chem., 2016, 55A, 413-422.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, et al. Gaussian 03, Revision C.02, Gaussian, Inc, 2004.
- R. Ghiasi, M. Z. Fashami, Tautomeric transformations and reactivity of isoindole and sila-indole: A computational study, J. Theor. Comput. Chem., 2014, 13, 1450041.
- A. E. Reed, L. a. Curtiss, F. Weinhold, Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint, Chem. Rev., 1988, 88, 899-926.
- H. Bernhard Schlegel, Optimization of equilibrium geometries and transition structures, J. Comput. Chem., 1982, 3, 214-218.
- J. Tomasi, M. Persico, Molecular-interactions in solution - An overview of methods based on continuous distributions of the solvent, Chem. Rev., 1994, 94, 2027-2094.
- E. Cances, B. Mennucci, J. Tomasi, A new integral equation formalism for the polarizable continuum model: Theoretical background and applications to isotropic and anisotropic dielectrics, J. Chem. Phys., 1997, 107, 3032-3041.
- R. G. Parr, L. V. Szentpaly, S. Liu, Electrophilicity Index, J. Am. Chem. Soc., 1999, 121, 1922-1924.
- R. G. Parr, R. G. Pearson, Absolute hardness: companion parameter to absolute electronegativity, J. Am. Chem. Soc., 1983, 105, 7512-7516.
- R. G. Parr and W. Yang, Density Functional Theory of Atoms and Molecules, Oxford University Press, New York, 1989, pp. 333.
- L. R. Domingo, E. Chamorro, P. Pérez, Understanding the Reactivity of Captodative Ethylenes in Polar Cycloaddition Reactions. A Theoretical Study, J. Org. Chem., 2008, 73, 4615-4624.
- L. R. Domingo, P. Pérez, The nucleophilicity N index in organic chemistry, Org. Biomol. Chem., 2011, 9, 7168-7175.
- W. Kohn, L. Sham, Self-Consistent Equations Including Exchange and Correlation Effects, J. Phys. Rev., 1965, 140, 1133-1138.
- R. Huisgen, P. Pöchlauer, G. Mlostoń, K. Polborn, Reactions of di(tert-butyl) diazomethane with acceptor-substituted ethylenes, Helv. Chim. Acta., 2007, 90, 983-998.
- S. A. Siadati, An example of a stepwise mechanism for the catalyst-free 1,3-dipolar cycloaddition between a nitrile oxide and an electron rich alkene, Tetrahedron Lett., 2015, 56, 4857-4863.
- L.R. Domingo, J.A. Saez, J.A. Joule, L. Rhyman, P, Ramasami, A DFT Study of the [3 + 2] versus [4 + 2] Cycloaddition Reactions of 1,5,6-Trimethylpyrazinium-3-olate with Methyl Methacrylate, J. Org. Chem., 2013, 78, 1621-1629.
- R. Jasiński, K. Mróz, A. Kącka, Experimental and Theoretical DFT Study on Synthesis of Sterically Crowded 2,3,3(4)5-Tetrasubstituted-4-nitroisoxazolidines via 1,3-Dipolar Cycloaddition Reactions Between Ketonitrones and Conjugated Nitroalkenes, J. Heterocycl. Chem., 2016, 53, 1424-1429.
- X. Li, D. Wei, Z. Li, Theoretical study on DBU-catalyzed insertion of isatins into aryl difluoronitromethyl ketones: A case for predicting chemoselectivity using electrophilic parr function, ACS Omega, 2017, 2, 7029-7038.
- E. Chamorro, P. Pérez, L.R. Domingo, On the nature of Parr functions to predict the most reactive sites along with organic polar reactions, Chem. Phys. Lett., 2013, 582, 141-143.
- C. Morell, J.L. Gázquez, A. Vela, F. Guégan, H. Chermette, Revisiting electroaccepting and electrodonating powers: proposals for local electrophilicity and local nucleophilicity descriptors, Phys. Chem. Chem. Phys., 2014, 16, 26832-26842.
- R. Jasiński, M. Ziółkowska, O.M. Demchuk, A. Maziarka, Regio- and stereoselectivity of polar [2+3] cycloaddition reactions between (Z)-C-(3,4,5-trimethoxyphenyl)-N-methylnitrone and selected (E)-2-substituted nitroethenes, Cent. Eur. J. Chem., 2014, 12, 586-593.
- A. K. Nacereddine, H. Layeb, F. Chafaa, A. Djerourou, L. R. Domingo, A DFT study of the role of the Lewis acid catalysts in the [3+ 2] cycloaddition reaction of the electrophilic nitrone isomer of methyl glyoxylate oxime with nucleophilic cyclopentene, RSC Adv., 2015, 5, 64098-64105.
- Z. Lakbaibi, A. Jaafar, H. Ben EL Ayouchia,
M. Tabyaoui, A. Boussaoud, Reactivity and mechanism of nucleophilic addition reaction of amine with alkene: A systematic DFT study J. Mediterranean of Chemistry, 2019, 8(1), 25-29.
- A. Benallou, Z. Lakbaibi, H. Garmes, H. El Alaoui EL Abdallaoui, The role of the polarity on the mechanism and selectivity in the [3+2] cycloaddition reaction between CF3-ynone ylide and azide group: A quantum chemical investigation J. Fluorine. Chem, 2019, 219, 79-91.
- L. R. Domingo, A new C–C bond formation model based on the quantum chemical topology of electron density, RSC Adv., 2014, 4,
-32428.
DOI: http://dx.doi.org/10.13171/mjc92190919645aoa/hbe
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
