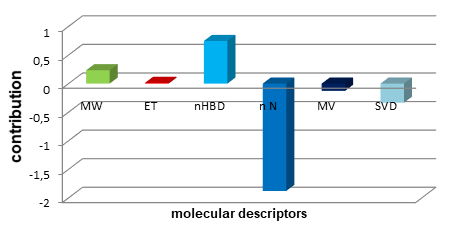
QSAR studies combined with DFT-calculations and Molecular docking of polyamine-sensitive inhibitors of the NMDA receptor
Abstract
Full Text:
PDFReferences
- S.F. Traynelis, L.P. Wollmuth, C.J. Mcbain, J. M. Chris, S.M. Frank, M.V. Katie, K.O. Kevin, B.H. Kasper, Y. Hongjie, J.M. Scott, D. Ray, Glutamate receptor ion channels: structure, regulation, and function, Pharmacol. Rev., 2010, 62, 405-496.
- S. Cull-Candy, S. Brickley, M. Farrant, NMDA receptor subunits: diversity, development and disease, Curr.Opin.Neurobiol., 2001, 11,
-335.
- S.F. Traynelis, M. Hartley, S.F. Heinemann, Control of proton sensitivity of the NMDA receptor by RNA splicing and polyamines, Sci., 1995, 26, 873-876.
- K. Williams, C. Romano, P.B. Molinoff, Effects of polyamines on the binding of [3H]MK-801 to the N-methyl-D-aspartate receptor: pharmacological evidence for the existence of a polyamine recognition site, Mol.Pharmacol., 1989, 36, 575-581.
- P. Thomas, S. Oliver, N. Daniela, R. Patrick, L.B. Michael, R.N. Christian, New polyamine-sensitive inhibitors of the NMDA receptor: Syntheses and pharmacological evaluation, Eur. Jour. Medici. Chem., 2007, 42, 175-197.
- H. Hadni, M. Mazigh, E. Charif, A. Bouayad, M. Elhallaoui, Molecular Modeling of Antimalarial Agents by 3D-QSAR Study and Molecular Docking of Two Hybrids 4-amino-quinoline-1,3,5-triazine and 4-amino-quinoline-oxalamide Derivatives with the Receptor Protein in Its Both Wild and Mutant Types, Biochem. Res. Int., 2018, 1-15.
- M.J. Frish, G. W. Turcks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. J. A. Montgomery, T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. lyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G.A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. Pople A. Revision D.01, Gaussian, Inc., Wallingford, CT, 2004.
- R. Parr, W. Yang, Density Functional Theory of Atoms and Molecules, Oxf. Uni. Pre., New York, 1989.
- A.D. Becke, ‹‹Density-Functional thermochemistry. III. The rol of exact exchange››, J. Chem. Phys., 1993, 98, 5648
- ACD/ChemSketch Version 4.5 for Microsoft Windows User's Guide.
- G. Saeed, G. Saeed, S. Ali, E.N. Heshmatollah, 2D-QSAR study of some 2,5-diaminobenzo-phenone farnesyltransferase inhibitors by different chemometric methods, ex.jour., 2015, 14, 484-495.
- S. Mbarki, M. El Hallaoui, 3D-QSAR models to predict antimoebic activities of the cyclised pyrazolines and 2-(quinolin-8-yloxy) acetohydrazones, res.Jour. Phar. Bio. Chem. Sci., 2014, 5, 73-83.
- S. So, G. Richards, Application of Neural Networks: Quantitative Structure-Activity Relationships of the Derivatives of 2,4-Diamino-5- (substituted-benzyl) pyrimidines as DHFR Inhibitors. J. Med. Chem., 1992, 35, 3201-3207.
- H. Hadni, M. Mazigh, M. Elhallaoui, QSAR and Molecular docking studies of 4-anilinoquinoline-triazine hybrids as pf-DHFR inhibitors, Mediterr. J. Chem., 2019, 8, 84-93.
- G. Morris, R. Huey, AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility, J. Comput. Chem., 2009, 30, 2785-2791.
- Discovery Studio Visualizer v4.1.0.14169 Copyright © 2005-14, Accelrys Software Inc.
- J. Rachline, F. Perin-Dureau, A.L. Goff, J. Neyton, P. Paoletti, The Micromolar Zinc-Binding Domain on the NMDA Receptor Subunit NR2B, Jour. Neu., 2005, 12, 308-317.
- R.N. Rama, Molecular Modeling: A Powerful Tool for Drug Design and Molecular Docking, Res., 2004, 9, 51-60.
DOI: http://dx.doi.org/10.13171/mjc93190924930hh
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
