Chemical preparation, thermal behavior and infrared studies of the new cyclotriphosphate tetrahydrate of manganese and distrontium, MnSr2(P3O9)2.4H2O
Abstract
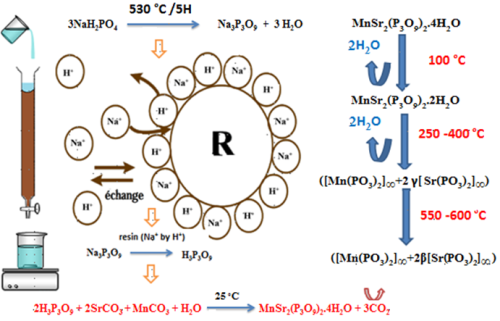
A new cyclotriphosphate tetrahydrate of manganese and distrontium, MnSr2(P3O9)2.4H2O, was obtained as a polycrystalline sample by the method of ion-exchange resin. The cyclotriphosphate studied in this work is the second one having the formula MnSr2(P3O9)2.4H2O after BaZn2(P3O9)2.10H2O. Its crystallographic characterization, infrared studies and thermal behavior are given. The studied phosphate crystallizes in the triclinic system, P-1, with the following unit cell parameters : a = 6,653(1)Å, b = 7,110(1)Å, c = 5,123(1)Å,
a = 103,37(2)°, b = 95,81(2)°, g = 93,04(2)° and Z = 1. The intermediate products of dehydration of MnSr2(P3O9)2.4H2O are MnSr2(P3O9)2.2H2O, mixture of ([Mn(PO3)2]∞ + 2g [Sr(PO3)2]∞) and mixture of ([Mn(PO3)2]∞ + 2b[Sr(PO3)2]∞). The thermal behavior of the studied condensed phosphate has been investigated and interpreted by IR absorption and X-ray diffraction experiments. The title compound can be used as catalyst, ion exchanger, the reactant in ionic conditions, intercalation reactions, superphosphate fertilizer and as an inorganic pigment in ceramics.
Full Text:
PDFReferences
- F. Álvaro, R.A. Johnstone, High surface area Pd, Pt and Ni ion-exchanged Zr, Ti and Sn (IV) phosphates and their application to selective heterogeneous catalytic hydrogenation of alkenes, Journal of Molecular Catalysis A: Chemical, 2008,280 (1-2),131-41. DOI:10.1016/j.molcata.2007.10.031
- A. Durif, M.T. Averbuch-Pouchot, J.C. Guitel, Crystalline structure of barium-zinc trimetaphosphatedecahydrate: Ba2Zn (P3O9) 2.10H2O, Acta Crystallographica Section B,1975, 31 (11), 2680-2682. DOI:10.1107/s056774087500845x.
- M. Tridane, S. Belaaouad, K. Sbai, Chemical preparations and crystal data for eight new condensed phosphates, Solid-state sciences, 2000, 7, 701-704. DOI:10.1016/S1293-2558(00)01081-5.
- K. Sbai, M. Tridane, A. Abouimrane, S. Belaaouad, A. Cherqaoui, Chemical preparation and crystal data for two new cyclotriphosphates MIIK4(P3O9)2 (M II= Co2+ and Mn2+), Powder Diffraction, 2003,18(3),224-6. DOI:10.1007/s100190000057
- S. Belaaouad, K. Sbai, Chemical preparation and crystal data for two new condensed phosphates, Powder Diffraction, 2002,17(1),23-4. DOI:10.1154/1.1420710
- A. Boultif and D. Louër, Powder pattern indexing with the dichotomy method, Journal of Applied Crystallography, 2004, 37, 724-731. DOI:10.1107/S0021889804014876.
- S. Zerraf, M. Belhabra, A. Kheireddine, R. Lamsatfi, M.Tridane, H. Moutaabbid, B. Baptiste, M. Moutaabbid and S.Belaaouad, Reinvestigation of the crystal structure of barium cesium cyclotriphosphatedihydrate and vibrational study, Phosphorus, Sulfur, and Silicon and the Related Elements, 2017, 192, 1286-1293. DOI:10.1080/10426507.2017.1333507.
- S. Zerraf, M. Tridane, S. Belaaouad, Data of infrared vibration spectroscopy of cyclotriphosphates, Data in Brief, 2019, 25,104075. DOI: 10.1016/j.dib.2019.104075.
- M. Belhabra, S. Zerraf, A. Kheireddine A. Altomare, M. Tridane, A. Ouasri, M. Radid and S. Belaaouad, Structural and vibrational study of diphenylhydrazinedihydrogenophosphate single crystal (C6H9N2)2H2P2O7(DPHDP), Chemical Data Collections, 2018,13, 73-83. DOI:10.1016/j.cdc.2018.01.002.
- A. Atibi, K. El Kababi, S. Zerraf, M. Tridane, S. Belaaouad, Chemical preparation crystal structure and vibrational study of a new dihydrogenotriphosphatetrihydrate of 4-aminobenzoic acid fertilizer type NP, Journal of Coordination Chemistry, 2018, 71, 3510-3520. DOI: 10.1080/00958972.2018.1528579.
- M. Belhabra, I. Fahim, A. Atibi, K. El Kababi, A. Ouasri, S. Zerraf, M. Tridane, M. Radid, S. Belaaouad, Vibrational study and thermal behavior of dihydrogenotriphosphatetrihydrate of 4-aminobenzoic acid and its anhydrous new form fertilizer type NP, Mediterranean Journal of Chemistry, 2019, 270-82. DOI:10.13171/mjc841905308mb.
- M. Graia, A. Driss, T. Jouini, Polyphosphates de strontium Sr(PO3)2 formes β et γ. Acta Crystallographica Section C: Communications en structure crystalline, 1999, 55, 1395-8. DOI:10.1107/S0108270199003637
- HA. Höppe, The synthesis, crystal structure and vibrational spectra of α-Sr (PO3)2 containing an unusual catena-polyphosphate helix. Solid-state sciences, 2005, 7(10), 1209-15. DOI:10.1016/j.solidstatesciences.2005.06.014.
- M.P. Gashti, M. Stir, J. Hullinger, Growth of strontium hydrogen phosphate/gelatin composites: a biomimetic approach, New journal of chemistry, 2016, 40(6), 5495-500. DOI: 10.1039/c5nj03575a.
- M. Weil, M. Uchberger, J. Schmedtauf der Guenne, J. Weber, Synthesis, Crystal Structure, and Characterization (Vibrational and Solid-State 31P MAS NMR Spectroscopy) of the High-Temperature Modification of Calcium catena-Polyphosphate (V), Chemistry of Materials, 2007,19(21), 5067-73. DOI:10.1021/cm071239i.
- S. Belaaouad, M. Tridane, H. Chennak, R. Tamani, A. Kenz, M. Moutaabbid, Chemical preparation, thermal behavior, kinetic and infrared studies and quantum chemical calculations of Ca3(P3O9)2.10H2O, Phosphorus Research Bulletin, 2007,21,60-70. DOI: 10.3363/prb.21.60.
-S. Belaaouad, Y. Lahrir, S.Sarhane, M. Tridane, Chemical preparation, thermal behavior, kinetic and infrared studies and quantum chemical calculations of Ba3 (P3O9)2.6H2O, Phosphorus Research Bulletin, 2009, 23, 67-75. DOI:10.3363/prb.23.67.
DOI: http://dx.doi.org/10.13171/mjc941911141082sem
Refbacks
- There are currently no refbacks.
Copyright (c) 2019 Mediterranean Journal of Chemistry
