Chemical composition and biological evaluation of essential oils from some Moroccan plants
Abstract
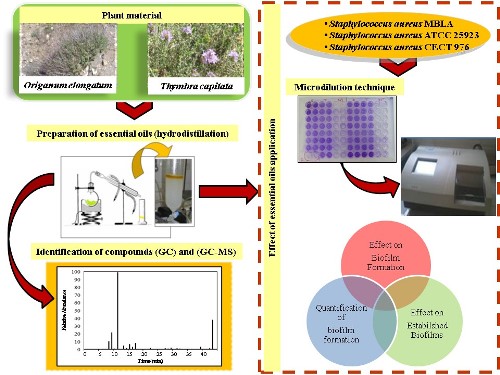
This study describes the chemical composition and biological activities of essential oils of the Moroccan plants Thymbra capitata (L.) Cav. and Origanum elongatum (Bonnet) Emb. & Maire on biofilms of Staphylococcus aureus strains. The qualitative and quantitative composition of the two essential oils was analyzed by gas chromatography and gas chromatography-mass spectrometry. The Minimum Inhibitory Concentration was determined by the broth microdilution method. The results showed that essential oils of Thymbra capitata and Origanum elongatum inhibit biofilm formation in biofilm inhibitory concentrations (0.5 and 2% v/v) and eradicate biofilm at biofilm eradication concentrations (1 and> 4% v/v), specific to each essential oil. Values were twofold or fourfold higher than the concentration required to inhibit or eradicate planktonic bacteria growth. The use of essential oils of Thymbra capitata and Origanum elongatum as natural antimicrobial agents may effectively inhibit the growth of biofilms of medical relevance. Results indicated that essential oils of Thymbra capitata and Origanum elongatum might be a potential alternative against Staphylococcus resistant strains.
Full Text:
PDFReferences
- J.W. Costerton, P.S. Stewart, E.P. Greenberg, Bacterial biofilms: a common cause of persistent infections. Science, 1999, 284, 1318-1322.
- A.L. Spoering, K. Lewis, Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials, J. Bacteriol., 2001, 183, 6746-6751.
- A.S. Fauci, E. Braunwald, K.J. Issekbacher, J.D. Wilson, J.B. Martin, D.L. Kasper, S.L. Hauser, D.L. Longo, Editors. Harrison’s Principles of Internal Medicine, McGraw-Hill New York, 1998.
- M. Chen, J. Cheng, Q. Wu, J. Zhang, Y. Chen, H. Zeng, Q. Ye, S. Wu, S. Cai, J. Wang, Y. Ding, Prevalence, potential virulence, and genetic diversity of Listeria monocytogenes isolates from edible mushrooms in Chinese markets, Front. Microbiol., 2018, 9, 1711.
- M. Simões, L.C. Simões, M.J. Vieira, A review of current and emergent biofilm control strategies, Food Sci. Technol., 2010, 43, 573-583.
- V. Sharma, S. Sharma, D.K. Dahiya, A.Khan, M. Mathur, A. Sharma, Coagulase gene polymorphism, enterotoxigenecity, biofilm production, and antibiotic resistance in Staphylococcus aureus isolated from bovine raw milk in northwest India, Ann Clin. Microbiol. Antimicrob. 2017, 16, 65.
- K.K. Kumarasamy, M.A. Toleman, T.R. Walsh, J. Bagaria, F. Butt, R. Balakrishnan, U. Chaudhary, M. Doumith, C.G. Giske, S. Irfan, P. Krishnan, A.V. Kumar, S. Maharjan, S. Mushtaq, T. Noorie, D.L. Paterson,
A. Pearson, C. Perry, R. Pike, B. Rao, U. Ray, J.B. Sarma, M. Sharma, E. Sheridan, M.A. Thirunarayan, J. Turton, S. Upadhyay, M. Warner, W. Welfare, D.M. Livermore, N. Woodford, Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study, Lancet Infect. Dis., 2010, 10, 597-602.
- N. El Moussaoui, G. Sanchez, K. El Ouardy, M. Idaomar, A. Ibn Mansour, J. Abrini, R. Aznar, Antibacterial and antiviral activities of essential oils of northern Moroccan plants, Br. Biotechnol. J., 2013, 3, 318-331.
- J. Bellakhdar, Ibis Press. Paris, 1997, 764.
- A. Benabid, Édition Ibis press, Paris, France, 2000, 236.
- K. Bakhy, O. Benlhabib, C. Al Faiz, A. Bighelli, J. Casanova, F. Tomi, Wild Thymbra capitata from Western Rif (Morocco): essential oil composition, chemical homogeneity and yield variability, Nat. Prod. Commun., 2013, 8, 1155-1158.
- A. Nostro, A. Sudano Roccaro, G. Bisignano, A. Marino, M.A. Cannatelli, F.C. Pizzimenti, P.L. Cioni, F. Procopio, A.R. Blanco, Effects of oregano, carvacrol and thymol on Staphylococcus aureus and Staphylococcus epidermidis biofilms, J. Med. Microbiol., 2007, 56, 519-523.
- A. Bouyahya, J. Abrini, N. Dakka, Y. Bakri, Essential oils of Origanum compactum increase membrane permeability, disturb cell membrane integrity, and suppress quorum-sensing phenotype in bacteria, J. Pharm. Anal., 2019, 9, 301-311.
- S. Charfi, N Boujida., N. El Moussaoui,J. Abrini, N. Skali Senhaji, Thymbra capitata Essential Oil Use to Preserve Physicochemical and Microbiological Qualities of Pomegranate Juice, Food Sc. Technol. Res., 2019, 2, 257-263.
- R. F. Cockerill, M.A. Wikler, J. Alder, M.N. Dudley, M. Eliopoulos, M. J. Ferraro, J. Hardy, D.W. Hecht, J. A. Hindler, Clinical and Laboratory Standards Institute. Method for dilution antimicrobial susceptibility tests for bacteria that grow aerobically approved standard-seventh edition. Clinical and Laboratory Standards Institute document M7-A7, 2006, 26, 1-49.
- G.D. Christensen, W.A. Simpson, J.J. Younger, L.M. Baddour, F.F. Barrett, D.M. Melton, D.H. Beachey, Adherence of coagulase-negative staphylococci to plastic tissue culture plates: a quantitative model for the adherence of staphylococci to medical devices, J. Clin. Microbiol., 1985, 22, 996-1006.
- T.J. Kärpänen, T. Worthington, E.R. Hendry, Antimicrobial efficacy of chlorhexidine digluconate alone and in combination with eucalyptus oil, tea tree oil and thymol against planktonic and biofilm cultures of Staphylococcus epidermidis, J. Antimicrob. Chemother., 2008, 62, 1031-1036.
- S.E. Cramton, C. Gerke, N.F. Schnell, W.W. Nichols, F. Götz, The intercellular adhesion (ica) locus in Staphylococcus aureus and is required for biofilm formation, Infect. Immun., 1999, 67, 5427-5433.
- B.J. Salas, R.T. Tellez, P.J.M. Alonso, V.F.M. Pardo, M. Capdevila, G. Rodriguez, Chemical composition and antioxidant activity of the essential oil of Thymbra capitata (L.) Cav. in Spain, Acta Bot. Gallica., 2010, 157, 55-63.
- A. Moukhles, A. Ibn Mansour, A. Ellaghdach, J. Abrini, Chemical composition and in vitro antibacterial activity of the pure essential oils and essential oils extracted from their corresponding hydrolats from different wild varieties of Moroccan thyme, J. Mater. Environ. Sci., 2018, 9, 1, 235-244.
- H. Oualili, R. Nmila, F. Chibi, M. Lasky, A. Mricha, H. Rachid, Chemical composition and antioxidant activity of Origanum elongatum essential oil, Phcog. Res., 2019,11, 283-9.
- S. Boukhira, F. Bousta, S. Moularat, A. Abdellaoui, Z. Benziane Ouaritini, D. Bousta, Evaluation of the Preservative Properties of Origanum elongatum Essential Oil in a Topically Applied Formulation Under a Challenge Test, Phytothérapie, 2018, 1-7.
- K. Lewis, Persistor Cells, and the Riddle of Biofilm Survival, Bioch. (Moscow), 2004, 70, 267-274.
- P. Cos, K. Toté, T. Horemans, L. Maes, Biofilms: an extra hurdle for effective antimicrobial therapy, Curr. Pharm. Design., 2010, 16,
-95.
- I. Kolouchová, O. Maťátková, M. Paldrychová, Z. Kodeš, E. Kvasničková, K. Sigler, A. Čejková, J. Šmidrkal, K. Demnerová, J. Masák, Resveratrol, pterostilbene, and baicalein: plant-derived anti-biofilm agents, Folia Microbiol., 2018, 63, 3, 261-272.
- T. Coenye, H.J. Nelis, In vitro and in vivo model systems to study microbial biofilm formation, J. Microbiol. Methods, 2010, 83, 89-105.
- H. Ceri, M.E. Olson, C. Stremick, R.R. Read,
D. Morck, A. Buret, The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms, J. Clin. Microbiol., 1999, 37, 1771-1776.
- P.S. Stewart, J.W. Costerton, Antibiotic resistance of bacteria in biofilms, Lancet, 2001, 358, 135-138.
- P. Gilbert, D.G. Allison, A.J. Mc Bain, Biofilms in vitro and in vivo: do singular mechanisms imply cross-resistance? Symp, Ser. Soc. Appl. Microbiol., 2002, 92, 98-110.
- K. Poole, Pseudomonas aeruginosa: resistance to the max, Front. Microbiol., 2011, 2, 65.
- M.J. Ashby, J.E. Neale, S.J. Knott, I.A. Critchley, Effect of antibiotics on non-growing cells of biofilms of Escherichia coli, J. Antimicrob. Chemother., 1994, 33, 443-452.
- K. Lewis, Persister cells, dormancy, and infectious disease, Nat. Rev. Microbiol., 2007, 5, 48-56.
- J.R. Knowles, S. Roller, D.B. Murray, A.S. Naidu Kong, Antimicrobial action of carvacrol at different stages of dual-species biofilm development by Staphylococcus aureus and Salmonella typhimurium, Appl. Environ. Microbiol., 2005, 71, 797-803.
- R.J. Lambert, P.N. Skandamis, P.J. Coote, G.J. Nychas, A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol, J. Appl. Microbiol., 2001, 91, 453-462.
- B.J. Juven, J. Kanner, F. Schved, H. Weisslowicz, Factors that interact with the antibacterial action of thyme essential oil and its active constituents, J. Appl. Bacteriol., 1994, 76, 626-631.
- F.M. Schempp, K.E. Hofmann, J. Mi, F. Kirchner, A. Meffert, H. Schewe, J. Schrader, M. Buchhaupt, Investigation of monoterpenoid resistance mechanisms in Pseudomonas putida and their consequences for biotransformations, Appl. Microbiol. Biotechnol., 2020, 1-15.
- A. Nostro, M.A. Cannatelli, I. Morelli, A.D. Musolino, F. Scuderi, F. Pizzimenti, V. Alonzo, Efficiency of Calaminthaofficinalis essential oil as a preservative in two topical product types, J. App.l Microbiol., 2004, 97, 395-401.
- K.R. Zodrow, J.D. Schiffman, M. Elimelech, Biodegradable Polymer (PLGA) Coatings Featuring Cinnamaldehyde and Carvacrol Mitigate Biofilm Formation, Langmuir, 2012, 28, 13993-13999.
DOI: http://dx.doi.org/10.13171/mjc10702007281485hd
Refbacks
- There are currently no refbacks.
Copyright (c) 2020 Mediterranean Journal of Chemistry
