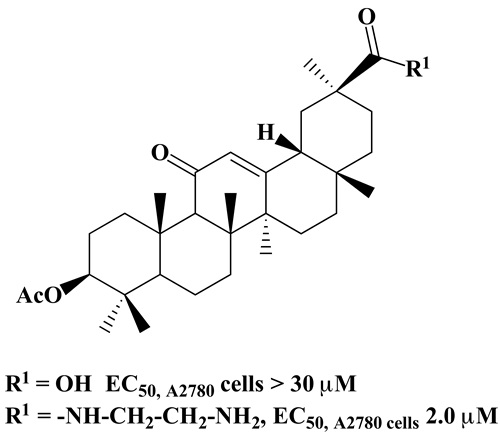
Glycyrrhetinic amides and their cytotoxicity
Abstract
Full Text:
PDFReferences
- X. Feng, L. Ding, F. Qiu, Potential drug interactions associated with glycyrrhizin and glycyrrhetinic acid, Drug Metab. Rev., 2015, 47, 229-238.
- H. Hussain, I.R. Green, U. Shamraiz, M. Saleem, A. Badshah, G. Abbas, N. Ur Rehman, M. Irshad, Therapeutic potential of glycyrrhetinic acids: a patent review (2010-2017), Expert Opin. Ther. Pat., 2018, 28, 383-398.
- X. Li, R. Sun, R. Liu, Natural products in licorice for the therapy of liver diseases: Progress and future opportunities, Pharmacol. Res., 2019, 144, 210-226.
- S.A. Richard, Exploring the pivotal immunomodulatory and anti-inflammatory potentials of glycyrrhizic and glycyrrhetinic acids, Mediators Inflammation, 2021.
- A. Roohbakhsh, M. Iranshahy, M. Iranshahi, Glycyrrhetinic Acid and Its Derivatives: Anti-Cancer and Cancer Chemopreventive Properties, Mechanisms of Action and Structure- Cytotoxic Activity Relationship, Curr. Med. Chem., 2016, 23, 498-517.
- H. Sharma, P. Kumar, R.R. Deshmukh, A. Bishayee, S. Kumar, Pentacyclic triterpenes: New tools to fight metabolic syndrome, Phytomedicine, 2018, 50, 166-177.
- Z.H. Tang, T. Li, Y.G. Tong, X.J. Chen, X.P. Chen, Y.T. Wang, J.J. Lu, A Systematic Review of the Anticancer Properties of Compounds Isolated from Licorice (Gancao), Planta Med., 2015, 81, 1670-1687.
- S. Wang, Y. Zhang, T. Zhang, J. Wang, W. Xu, Y. Zhang, Y. Luo, C. Jin, Advances in research on anti-cancer mechanism of 18β glycyrrhetinic acid, Med. Plant, 2019, 10, 10-12.
- R. Yang, L.q. Wang, B.c. Yuan, Y. Liu, The Pharmacological Activities of Licorice, Planta Med., 2015, 81, 1654-1669.
- R. Yang, B.C. Yuan, Y.S. Ma, S. Zhou, Y. Liu, The anti-inflammatory activity of licorice, a widely used Chinese herb, Pharm. Biol., 2017, 55, 5-18.
- B. Brandes, S. Hoenke, L. Fischer, R. Csuk, Design, synthesis and cytotoxicity of BODIPY FL labelled triterpenoids, Eur. J. Med. Chem., 2020, 185, 111858.
- M. Huang, P. Gong, Y. Wang, X. Xie, Z. Ma, Q. Xu, D. Liu, Y. Jing, L. Zhao, Synthesis and antitumor effects of novel 18β-glycyrrhetinic acid derivatives featuring an exocyclic α,β-unsaturated carbonyl moiety in ring A, Bioorg. Chem., 2020, 103, 104187.
- O. Kazakova, I. Smirnova, E. Tretyakova, R. Csuk, S. Hoenke, L. Fischer, Cytotoxic Potential of a-Azepanoand 3-Amino-3,4-SeCo-Triterpenoids, Int. J. Mol. Sci., 2021, 22, 1714.
- L. Li, S. Han, C. Yang, L. Liu, S. Zhao, X. Wang, B. Liu, H. Pan, Y. Liu, J. Pan, Y. Wang, J. Li, B. Jiang, R. Liu, X. Wang, X. Zhang, R. Zhang, Z.A. Qiao, Glycyrrhetinic acid modified MOFs for the treatment of liver cancer, Nanotechnology, 2020, 31, 325602.
- A.V. Markov, K.V. Odarenko, A.V. Sen'kova, O.V. Salomatina, N.F. Salakhutdinov, M.A. Zenkova, Cyano enone-bearing triterpenoid soloxolone methyl inhibits epithelial-mesenchymal transition of human lung adenocarcinoma cells in vitro and metastasis of murine melanoma in vivo, Molecules, 2020, 25, 5925.
- J. Shi, J. Li, J. Li, R. Li, X. Wu, F. Gao, L. Zou, W.W.S. Mak, C. Fu, J. Zhang, G.P.H. Leung, Synergistic breast cancer suppression efficacy of doxorubicin by combination with glycyrrhetinic acid as an angiogenesis inhibitor, Phytomedicine, 2021, 81, 153408.
- R. Wang, W. Yang, Y. Fan, W. Dehaen, Y. Li, H. Li, W. Wang, Q. Zheng, Q. Huai, Design and synthesis of the novel oleanolic acid-cinnamic acid ester derivatives and glycyrrhetinic acid-cinnamic acid ester derivatives with cytotoxic properties, Bioorg. Chem., 2019, 88, 102951.
- R.K. Wolfram, L. Fischer, R. Kluge, D. Stroehl, A. Al-Harrasi, R. Csuk, Homopiperazine-rhodamine B adducts of triterpenoic acids are strong mitocans, Eur. J. Med. Chem., 2018, 155, 869-879.
- R.K. Wolfram, L. Heller, R. Csuk, Targeting mitochondria: Esters of rhodamine B with triterpenoids are mitocanic triggers of apoptosis, Eur. J. Med. Chem., 2018, 152, 21-30.
- Q.X. Zheng, R. Wang, Y. Xu, C.X. He, C.Y. Zhao, Z.F. Wang, R. Zhang, W. Dehaen, H.J. Li, Q.Y. Huai, Design, preparation and studies regarding cytotoxic properties of glycyrrhetinic acid derivatives, Biol. Pharm. Bull., 2020, 43, 102-109.
- R. Sczepek, C. Nitsche, L. Heller, B. Siewert, R. Schaefer, F. Flemming, C. Otgonbayar, R. Csuk, Synthesis and cytotoxic properties of alkynic triterpenoid Mannich compounds, Mediterr. J. Chem., 2015, 4, 126-137.
- B. Brandes, L. Koch, S. Hoenke, H.P. Deigner, R. Csuk, The presence of a cationic center is not alone decisive for the cytotoxicity of triterpene carboxylic acid amides, Steroids, 2020, 163, 108713.
- S. Friedrich, I. Serbian, S. Hoenke, R.K. Wolfram, R. Csuk, Synthesis and cytotoxic evaluation of malachite green derived oleanolic and ursolic acid piperazineamides, Med. Chem. Res., 2020, 29, 926-933.
- S. Hoenke, M.A. Christoph, S. Friedrich, N. Heise, B. Brandes, H.P. Deigner, A. Al-Harrasi, R. Csuk, The presence of a cyclohexyldiamine moiety confers cytotoxicity to pentacyclic triterpenoids, Molecules, 2021, 26, 2102.
- O. Kazakova, E. Tret'yakova, D. Baev, Evaluation of A-azepano-triterpenoids and related derivatives as antimicrobial and antiviral agents, J. Antibiot., 2021.
- O. Kraft, M. Kozubek, S. Hoenke, I. Serbian, D. Major, R. Csuk, Cytotoxic triterpenoid-safirinium conjugates target the endoplasmic reticulum, Eur. J. Med. Chem., 2021, 209, 112920.
- G.E. Conway, D. Zizyte, J.R.M. Mondala, Z. He, L. Lynam, M. Lecourt, C. Barcia, O. Howe, J.F. Curtin, Ursolic acid inhibits collective cell migration and promotes JNK-dependent lysosomal associated cell death in glioblastoma multiforme cells, Pharmaceuticals, 2021, 14, 91.
- E.F. da Silva, A.S. de Vargas, J.B. Willig, C.B. de Oliveira, A.R. Zimmer, D.A. Pilger, A. Buffon, S.C.B. Gnoatto, Synthesis and antileukemic activity of an ursolic acid derivative: A potential co-drug in combination with imatinib, Chem.-Biol. Interact., 2021, 344, 109535.
- R. Hu, J. Sang, W. Li, Y. Tian, M.F. Zou, G.H. Tang, S. Yin, Structurally diverse triterpenoids with cytotoxicity from Euphorbia hypericifolia, Fitoterapia, 2021, 151, 104888.
- A.Y. Spivak, R.R. Khalitova, R.R. Gubaidullin, D.A. Nedopekina, Synthesis and cytotoxic activity of monomeric and dimeric aminocarboxamides of betulinic and ursolic acids, Chem. Nat. Compd., 2021, 57, 123-132.
- M. Yang, C. Hu, Y. Cao, W. Liang, X. Yang, T. Xiao, Ursolic acid regulates cell cycle and proliferation in colon adenocarcinoma by suppressing cyclin B1, Front. Pharmacol., 2020, 11, 622212.
- T.Y. Zhang, C.S. Li, L.T. Cao, X.Q. Bai, D.H. Zhao, S.M. Sun, New ursolic acid derivatives bearing 1,2,3-triazole moieties: design, synthesis and anti-inflammatory activity in vitro and in vivo, Mol. Diversity, 2021.
- M. Kahnt, A. Loesche, I. Serbian, S. Hoenke, L. Fischer, A. Al-Harrasi, R. Csuk, The cytotoxicity of oleanane derived aminocarboxamides depends on their aminoalkyl substituents, Steroids, 2019, 149, 108422.
- M. Kahnt, J. Wiemann, L. Fischer, S. Sommerwerk, R. Csuk, Transformation of asiatic acid into a mitocanic, bimodal-acting rhodamine B conjugate of nanomolar cytotoxicity, Eur. J. Med. Chem., 2018, 159,
-148.
- K.W. Lu, M.D. Yang, S.F. Peng, J.C. Chen, P.Y. Chen, H.Y. Chen, T.J. Lu, F.S. Chueh, J.C. Lien, K.C. Lai, K.C. Liu, Y.Y. Tai, Maslinic acid induces DNA damage and impairs DNA repair in human cervical cancer HeLa cells, Anticancer Res., 2020, 40, 6869-6877.
- I.Z. Pavel, C. Danciu, C. Oprean, C.A. Dehelean, D. Muntean, R. Csuk, D.M. Muntean, In vitro evaluation of the antimicrobial ability and cytotoxicity on two melanoma cell lines of a benzylamide derivative of maslinic acid, Anal. Cell. Pathol., 2016.
- I. Serbian, B. Siewert, A. Al-Harrasi, R. Csuk, 2-O-(2-chlorobenzoyl) maslinic acid triggers apoptosis in A2780 human ovarian carcinoma cells, Eur. J. Med. Chem., 2019, 180, 457-464.
- S. Sommerwerk, L. Heller, C. Kerzig, A.E. Kramell, R. Csuk, Rhodamine B conjugates of triterpenoic acids are cytotoxic mitocans even at nanomolar concentrations, Eur. J. Med. Chem., 2017, 127, 1-9.
- S. Sommerwerk, L. Heller, J. Kuhfs, R. Csuk, Urea derivates of ursolic, oleanolic and maslinic acid induce apoptosis and are selective cytotoxic for several human tumor cell lines, Eur. J. Med. Chem., 2016, 119, 1-16.
- K. Vega-Granados, M. Medina-O'Donnell, F. Rivas, F.J. Reyes-Zurita, A. Martinez, L. Alvarez de Cienfuegos, J.A. Lupianez, A. Parra, Synthesis and Biological Activity of Triterpene-Coumarin Conjugates, J. Nat. Prod., 2021, 84, 1587-1597.
- U. Bildziukevich, Z. Ozdemir, Z. Wimmer, Recent achievements in medicinal and supramolecular chemistry of betulinic acid and its derivatives, Molecules, 2019, 24, 3546.
- S. Fulda, Betulinic acid: a natural product with anticancer activity, Mol. Nutr. Food Res., 2009, 53, 140-146.
- S. Fulda, G. Kroemer, Targeting mitochondrial apoptosis by betulinic acid in human cancers, Drug Discovery Today, 2009, 14, 885-890.
- M. Grymel, M. Zawojak, J. Adamek, Triphenylphosphonium Analogues of Betulin and Betulinic Acid with Biological Activity:
A Comprehensive Review, J. Nat. Prod., 2019, 82, 1719-1730.
- I. Mierina, R. Vilskersts, M. Turks, Delivery Systems for Birch-bark Triterpenoids and their Derivatives in Anticancer Research, Curr. Med. Chem., 2020, 27, 1308-1336.
- R. Mukherjee, V. Kumar, S.K. Srivastava, S.K. Agarwal, A.C. Burman, Betulinic acid derivatives as anticancer agents: structure-activity relationship, Anti-Cancer Agents Med. Chem., 2006, 6, 271-279.
- J. Sarek, M. Kvasnica, M. Vlk, D. Biedermann, in Pentacyclic triterpnes as promising agents in cancer, ed. by J.A. R. Salvador, Nova Science Publishers, Inc.: New York, 2010, 159-189.
- L. Tripathi, P. Kumar, R. Singh, A review on extraction, synthesis and anticancer activity of betulinic acid, Curr. Bioact. Compd., 2009, 5, 160-168.
- D.M. Zhang, H.G. Xu, L. Wang, Y.J. Li, P.H. Sun, X.M. Wu, G.J. Wang, W.M. Chen, W.C. Ye, Betulinic Acid and its Derivatives as Potential Antitumor Agents, Med. Res. Rev., 2015, 35, 1127-1155.
- I. Beseda, L. Czollner, P.S. Shah, R. Khunt, R. Gaware, P. Kosma, C. Stanetty, M.C. del Ruiz-Ruiz, H. Amer, K. Mereiter, T. Da Cunha, A. Odermatt, D. Classen-Houben, U. Jordis, Synthesis of glycyrrhetinic acid derivatives for the treatment of metabolic diseases, Bioorg. Med. Chem., 2010, 18, 433-454.
- E.E. Mikhlina, M.V. Rubtsov, Reaction of 3-quinuclidone with hydrazoic acid, Zh. Obshch. Khim., 1963, 33, 2167-2172.
- R. Csuk, S. Schwarz, B. Siewert, R. Kluge, D. Stroehl, Conversions at C-30 of Glycyrrhetinic Acid and Their Impact on Antitumor Activity, Arch. Pharm., 2012, 345, 223-230.
- G. Drefahl, S. Huneck, The preparation of acetylglycyrrhetinic acid and its Curtius degradation, Chem. Ber., 1961, 94, 2015-2018.
- D. Cai, Z. Zhang, Y. Meng, K. Zhu, L. Chen, C. Yu, C. Yu, Z. Fu, D. Yang, Y. Gong, Efficient synthesis of piperazinyl amides of 18β-glycyrrhetinic acid, Beilstein J. Org. Chem., 2020, 16, 798-808.
- K.A. Alibaeva, H.O. Kim, M.I. Goryaev, M.P. Irismetov, Triterpenoids. XXXI. Beckmann rearrangement of glycyrrhetic acid amides, Izv. Akad. Nauk Kaz. SSR, Ser. Khim., 1975, 25, 39-42.
- C.R. Montague, A. Fitzmaurice, B.M. Hover, N.A. Salazar, J.P. Fey, Screen for small molecules increasing the mitochondrial membrane potential, J. Biomol. Screening, 2014, 19, 387-398, 312.
- M.O. Radwan, M.A.H. Ismail, S. El-Mekkawy, N.S. M. Ismail, A.G. Hanna, Synthesis and biological activity of new 18β-glycyrrhetinic acid derivatives, Arabian J. Chem., 2016, 9, 390-399.
- C.H. Brieskorn, V. Beer, Formation of a tetraene from 18β-glycyrrhetic acid, Arch. Pharm., 1975, 308, 852-858.
- M.J. Kulshreshtha, R.P. Rastogi, 2α,3β-Dihydroxy triterpenoids. Partial syntheses of methyl alphitolate and methyl 2α-hydroxyursolate, Indian J. Chem., 1971, 9, 897-898.
- S. Rozen, I. Shahak, E.D. Bergmann, Derivatives of glycyrrhetic acid, Isr. J. Chem., 1971, 9, 185-189.
- H. Brieskorn, H. Sax, Synthesis of some derivatives of glycyrrhizic and glycyrrhetic acids, Arch. Pharm., 1970, 303, 905-912.
- P.D.G. Dean, T.G. Halsall, M.W. Whitehouse, Preparation of some derivatives of glycyrrhetic acid and oleanolic acid, J. Pharm. Pharmacol., 1967, 19, 682-689.
- R.K. Gayanov, H.O. Kim, M.P. Irismetov, M.I. Goryaev, Triterpenoids. XXXV. Reactions of glycyrrhetic acid amides, Zh. Obshch. Khim., 1978, 48, 920-923.
- C. Stanetty, L. Czollner, I. Koller, P. Shah, R. Gaware, T. Da Cunha, A. Odermatt, U. Jordis, P. Kosma, D. Classen-Houben, Synthesis of novel 3-amino and 29-hydroxamic acid derivatives of glycyrrhetinic acid as selective 11β-hydroxysteroid dehydrogenase 2 inhibitors, Bioorg. Med. Chem., 2010, 18, 7522-7541.
- M.W. Whitehouse, P.D.G. Dean, T.G. Halsall, Uncoupling of oxidative phosphorylation by glycyrrhetic acid, fusidic acid, and some related triterpenoid acids, J. Pharm. Pharmacol., 1967, 19, 533-544.
- L.W. Zou, Y.G. Li, P. Wang, K. Zhou, J. Hou, Q. Jin, D.C. Hao, G.B. Ge, L. Yang, Design, synthesis, and structure-activity relationship study of glycyrrhetinic acid derivatives as potent and selective inhibitors against human carboxylesterase 2, Eur. J. Med. Chem., 2016, 112, 280-288.
- A. Shukla, R. Tyagi, S. Meena, D. Datta, S.K. Srivastava, F. Khan, 2D- and 3D-QSAR modelling, molecular docking and in vitro evaluation studies on 18beta-glycyrrhetinic acid derivatives against triple-negative breast cancer cell line, J. Biomol. Struct. Dyn., 2020, 38, 168-185.
- R. Tyagi, S. Verma, S. Mishra, M. Srivastava, S. Alam, F. Khan, S.K. Srivastava, In Vitro and In Silico Studies of Glycyrrhetinic Acid Derivatives as Anti- Filarial Agents, Curr. Top. Med. Chem., 2019, 19, 1191-1200.
DOI: http://dx.doi.org/10.13171/mjc02110161595Cesuk
Refbacks
- There are currently no refbacks.
Copyright (c) 2021 Mediterranean Journal of Chemistry
