Inclusion Complex of Fexofenadine Hydrochloride with Cyclodextrins
Abstract
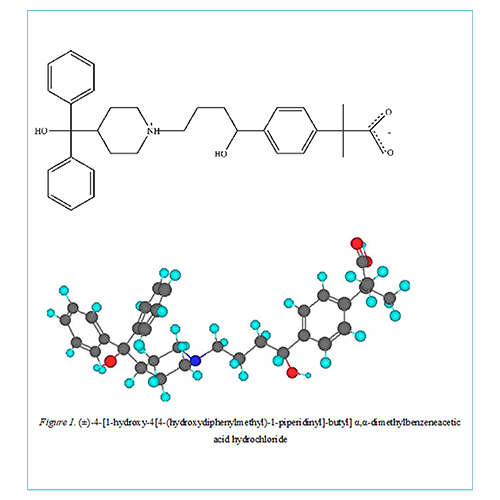
Fexofenadine hydrochloride (FFN), (±)-4-[1-hydroxy-4[4-(hydroxydiphenylmethyl)-1-piperidinyl]-butyl] α,α-dimethylbenzeneacetic acid hydrochloride, is a second-generation antihistamine that is used to treat allergies. The drug is highly hydrophobic and slightly soluble in water. Cyclodextrins are widely used to improve the physicochemical and pharmaceutical properties such as solubility, stability, and bioavailability of poorly soluble drug molecules.Cyclodextrins can molecularly encapsulate various drugs into their hydrophobic cavity without forming any covalent bonds. Cyclodextrin (CDs), especially ß-Cyclodextrin (ß-CD), are widely used in the pharmaceutical field due to its ability to stabilize drug molecules and taste masking purposes.
The phase solubility study was performed according to the method of Higuchi and Connors by adding the fexofenadine hydrochloride in excess to different concentrations of cyclodextrin solutions. Phase solubility study records show that the stability constant and complex stoichiometry of FFN-CD complexes increases linearly with CD concentration. Also, an increase in the concentration of β-cyclodextrin leads to an increase in the aqueous solubility of FFN. Complexes were analyzed by UV-VIS spectroscopy using the calibration curve of FFN. Also, UV-VIS spectra indicate a bathochromic shift which proves that complex formation has occurred.
Solid inclusion complexes of fexofenadine/β-cyclodextrin and its derivatives were prepared at the molar ratios of 1:1 by the physical mixing method. Characterization of the complexes was performed by using infrared spectroscopy.
Full Text:
PDFReferences
M. M. Heravi, V. Zadsirjan, Prescribed drugs containing nitrogen heterocycles: an overview, RSC Adv., 2020, 10, 44247-44311.
C. T. Abhiman, J. H. Anantrao, Phase solubility studies of glimepiride with ß-cyclodextrin and hydroxy propyl-ß-cyclodextrin in different pH, Indo American Journal of Pharmaceutical Research, 2017, 7, 604-612.
M. M. Al Omari, A. A. Badwan, M. B. Zughul, J. E. Davies, Fexofenadine/Cyclodextrin Inclusion Complexation: Phase Solubility, Thermodynamic, Physicochemical, and Computational Analysis Drug Development and Industrial Pharmacy, 2007, 33, 1205–1215.
N. P. Sapkal, V. A. Kilor, B. D. Shewale, K. P. Bhusari, A. S. Daud, Study of the Complexation Behaviour of Fexofenadine with β-
Cyclodextrin, Indian J Pharm Sci., 2010, 72(3), 318–323.
S. S. Jambhekar, P. Breen, Cyclodextrins in pharmaceutical formulations I: structure and physicochemical properties, formation of complexes, and types of complex; Drug Discovery Today, 2016, 21, 356-362.
H. Dodziuk, Molecules with holes-Cyclodextrins, Cyclodextrins and their complexes, 2006, 1-30.
P. Saokham, C. Muankaew, P. Jansook, T. Loftsson, Solubility of Cyclodextrins and Drug/Cyclodextrin Complexes, Journal Molecules, 2018, 23, 1161.
M. M. Al Omari, M. B. Zughul, E. D. Davies,
A. A. Badwan, Effect of buffer species on the complexation of basic drug terfenadine with β-cyclodextrin, Journal of Inclusion Phenomena Macrocyclic Chemistry., 2007, 58, 227-235.
N. Ojha, B. Prabhakar, Advances in Solubility Enhancement Techniques, Int. J. Pharm. Sci. Rev. Res., 2013, 21, 351-358.
M. M. Al Omari, A. A. Badwan, Fexofenadine/Cyclodextrin Inclusion Complexation: PhaseSolubility, Thermodynamic, Physicochemical and Computational Analysis, Drug Development and Industrial Pharmacy, 2007, 33, 1205-1215.
S. J. Pandya, T. Y. Pasha, A. Bhandari, J. K. Patel, T. Naitik, T. Upama, Physical Characterization and Enhance the Solubility of Fexofenadine-β-Cyclodextrin Inclusion Complexes, Current Pharma Research, 2011, 3, 260-266.
DOI: http://dx.doi.org/10.13171/mjc02112181597huremovic
Refbacks
- There are currently no refbacks.
Copyright (c) 2021 Mediterranean Journal of Chemistry
