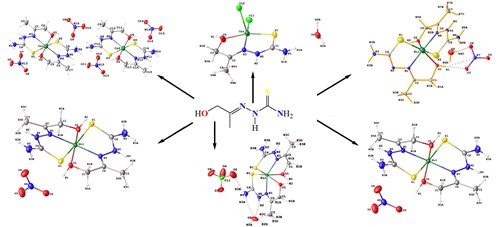
Transition metal complexes of 1-(1-hydroxypropan-2-ylidene) thiosemicarbazide (H2L): Synthesis, spectroscopic study, and X-ray diffraction structures
Abstract
Full Text:
PDFReferences
- O.A. El-Gammal, G.M.A. El-Reash, M.M. El-Gamil, Structural, spectral, pH-metric and biological studies on mercury (II), cadmium (II) and binuclear zinc (II) complexes of NS donor thiosemicarbazide ligand, Spectrochim. Acta, Part A, 2014, 123, 59–70. https://doi.org/10.1016/j.saa.2013.12.034.
- A. Basu, G. Das, Zn(II) and Hg(II) complexes of naphthalene based thiosemicarbazone: Structure and spectroscopic studies, Inorg. Chim. Acta, 2011, 372(1), 394–399. https://doi.org/10.1016/j.ica.2011.01.097.
- B. Wang, Z.-Y. Yang, M. Lü, J. Hai, Q. Wang, Z.-N. Chen, Synthesis, characterization, cytotoxic activity and DNA binding Ni(II) complex with the 6-hydroxy chromone-3-carbaldehyde thiosemicarbazone, J. Organomet. Chem., 2009, 694(25), 4069–4075. https://doi.org/10.1016/j.jorganchem.2009.08.024.
- H. Hosseinpoor, S.M. Farid, A. Iraji, M.S. Asgari, N. Edraki, S. Hosseini, A. Jamshidzadeh, B. Larijani, M. Attarroshan, S. Pirhadi, M. Mahdavi, M. Khoshneviszadeh, Anti-melanogenesis and anti-tyrosinase properties of aryl-substituted acetamides of phenoxy methyl triazole conjugated with thiosemicarbazide: Design, synthesis, and biological evaluations, Bioorg. Chem., 2021, 114, 104979. https://doi.org/10.1016/j.bioorg.2021.104979.
- F.S. Tokalı, P. Taslimi, H. Usanmaz, M. Karaman, K. Şendil, Synthesis, characterization, biological activity, and molecular docking studies of novel Schiff bases derived from thiosemicarbazide: Biochemical and computational approach, J. Mol. Struct., 2021, 1231, 129666. https://doi.org/10.1016/j.molstruc.2020.129666.
- Z. Bakherad, M. Mohammadi-Khanaposhtani, H. Sadeghi-Aliabadi, S. Rezaei, A. Fassihi, M. Bakherad, H. Rastegar, M. Biglar, L. Saghaie,
B. Larijani, M. Mahdavi, New thiosemicarbazide-1,2,3-triazole hybrids as potent α-glucosidase inhibitors: Design, synthesis, and biological evaluation, J. Mol. Struct., 2019, 1192, 192–200. https://doi.org/10.1016/j.molstruc.2019.04.082.
- H.A. Aboseada, M.M. Hassanien, I.H. El-Sayed, E.A. Saad, Schiff base 4-ethyl-1-(pyridin-2-yl) thiosemicarbazide up-regulates the antioxidant status and inhibits the progression of Ehrlich solid tumor in mice, Biochem. Biophys. Res. Commun., 2021, 573, 42–47. https://doi.org/10.1016/j.bbrc.2021.07.102.
- A.A. Altalhi, H.E. Hashem, N.A. Negm, E.A. Mohamed, E.M. Azmy, Synthesis, characterization, computational study, and screening of novel 1-phenyl-4-(2-phenylacetyl)-thiosemicarbazide derivatives for their antioxidant and antimicrobial activities. J. Mol. Liq., 2021, 333, 115977. https://doi.org/10.1016/j.molliq.2021.115977.
- L.S. Munaretto, M. Ferreira, D.P. Gouvêa, A.J. Bortoluzzi, L.S. Assunção, J. Inaba, T.B. Creczynski-Pasa, M.M. Sá, Synthesis of isothiosemicarbazones of potential antitumoral activity through a multicomponent reaction involving allylic bromides, carbonyl compounds and thiosemicarbazide, Tetrahedron, 2020, 76(23), 131231. https://doi.org/10.1016/j.tet.2020.131231.
- D.B. Patel, D.G. Darji, K.R. Patel, D.P. Rajani, S.D. Rajani, H.D. Patel, Synthesis of novel quinoline‐thiosemicarbazide hybrids and evaluation of their biological activities, molecular docking, molecular dynamics, pharmacophore model studies, and ADME‐Tox properties, J. Heterocycl. Chem., 2020, 57(3), 1183–1200. https://doi.org/10.1002/jhet.3855.
- D.B. Patel, K.D. Patel, N.P. Prajapati, K.R. Patel, D.P. Rajani, S.D. Rajani, N.S. Shah, D.D. Zala, H.D. Patel, Design, Synthesis, and Biological and In Silico Study of Fluorine‐Containing Quinoline Hybrid Thiosemicarbazide Analogues, J. Heterocycl. Chem., 2019, 56(8), 2235–2252. https://doi.org/10.1002/jhet.3617.
- B. Šarkanj, M. Molnar, M. Čačić, L. Gille, 4-Methyl-7-hydroxycoumarin antifungal and antioxidant activity enhancement by substitution with thiosemicarbazide and thiazolidinone moieties, Food Chem., 2013, 139(1), 488–495. https://doi.org/10.1016/j.foodchem.2013.01.027.
- T. Panneerselvam, J.R. Mandhadi, Microwave-assisted synthesis and antimicrobial evaluation of novel substituted thiosemicarbazide derivatives of pyrimidine, J. Heterocycl. Chem., 2020, 57(8), 3082–3088. https://doi.org/10.1002/jhet.4013.
- P.T. Acharya, Z.A. Bhavsar, D.J. Jethava, D.B. Patel, H.D. Patel, A review on the development of bio-active thiosemicarbazide derivatives: Recent advances, J. Mol. Struct., 2021, 1226, 129268. https://doi.org/10.1016/j.molstruc.2020.129268.
- M.S. Refat, A.A.M. Belal, I.I. El-Deen, N. Hassan, R. Zakaria, Synthesis, spectroscopic, thermal, and antimicrobial investigations of new mono and binuclear Cu(II), Co(II), Ni(II), and Zn(II) thiosemicarbazide complexes, J. Mol. Struct., 2020, 1218(5), 128516. https://doi.org/10.1016/j.molstruc.2020.128516.
- Y. Wang, H.-Q. Chang, W.-N. Wu, X.-J. Mao, X.-L. Zhao, Y. Yang, Z.-Q. Xu, Z.-H. Xu, L. Jia, A highly sensitive and selective colorimetric and off–on fluorescent chemosensor for Cu2+ based on rhodamine 6G hydrazide bearing thiosemicarbazide moiety, J. Photochem. Photobiol. A Chem., 2017, 335, 10-16. https://doi.org/10.1016/j.jphotochem.2016.11.003.
- V. Arion, K. Wieghardt, T. Weyhermueller, E. Bill, V. Leovac, A. Rufinska, Synthesis, structure, magnetism, and spectroscopic properties of some mono- and dinuclear nickel complexes containing noninnocent pentane-2,4-dione bis(S-alkylisothiosemicarbazonate)- derived ligands, Inorg. Chem., 1997, 36, 661–669. https://doi.org/10.1021/ic960802o.
- A. Fetoh, M.A. Mohammed, M.M, Youssef, G.M.A. El-Reash, Investigation (IR, UV-visible, fluorescence, X-ray diffraction and thermogravimetric) studies of Mn(II), Fe(III) and Cr(III) complexes of thiosemicarbazone derived from 4-pyridyl thiosemicarbazide and monosodium 5-sulfonatosalicylaldehyde and evaluation of their biological applications, J. Mol. Struct., 2023,1271, 134139. https://doi.org/10.1016/j.molstruc.2022.134139.
- N. Raman, A. Selvan, P. Manisankar, Spectral, magnetic, biocidal screening, DNA binding and photocleavage studies of mononuclear Cu(II) and Zn(II) metal complexes of tricoordinate heterocyclic Schiff base ligands of pyrazolone and semicarbazide/thiosemicarbazide based derivatives, Spectrochim. Acta, Part A, 2010, 76(2), 161–173. https://doi.org/10.1016/j.saa.2010.03.007.
- S. Chandra, X. Sangeetika, EPR, magnetic and spectral studies of copper(II) and nickel(II) complexes of Schiff base macrocyclic ligand derived from thiosemicarbazide and glyoxal. Spectrochim, Acta, Part A, 60(1), 2004, 147–153. https://doi.org/10.1016/S1386-1425(03)00220-8.
- A. Shaikh, P. Mukherjee, S. Ta, A. Bhattacharyya,
A. Ghosh, D. Das, Oxidative cyclization of thiosemicarbazide: a chemodosimetric approach for the highly selective fluorescence detection of cerium(IV), New J. Chem., 2020, 44(22), 9452–9455. https://doi.org/10.1039/D0NJ01100B.
- Y. Wang, H.-Q. Chang, W.-N. Wu, X.-J. Mao, X.-L. Zhao, Y. Yang, Z.-Q. Xu, Z.-H. Xu, L. Jia, A highly sensitive and selective colorimetric and off–on fluorescent chemosensor for Cu2+ based on rhodamine 6G hydrazide bearing thiosemicarbazide moiety, J. Photochem. Photobiol., A, 2017, 335, 10–16. https://doi.org/10.1016/j.jphotochem.2016.11.003.
- S. Angupillai, J.-Y. Hwang, J.-Y. Lee, B.A. Rao, Y.-A. Son, Efficient rhodamine-thiosemicarbazide-based colorimetric/fluorescent ‘turn-on’ chemodosimeters for the detection of Hg2+ in aqueous samples, Sens. Actuators, B, 2015, 214, 101–110. https://doi.org/10.1016/j.snb.2015.02.126.
- M. Salavati-Niasari, Host (nanocage of zeolite–Y)/guest manganese(II), cobalt(II), nickel(II) and copper(II) complexes of 12-membered macrocyclic Schiff-base ligand derived from thiosemicarbazide and glyoxal) nanocomposite materials: Synthesis, characterization and catalytic oxidation of cyclohexene, J. Mol. Catal. A: Chem., 2008, 283(1), 120–128. https://doi.org/10.1016/j.molcata.2007.12.015.
- B. Pouramiri, E.T. Kermani, Lanthanum(III) chloride/chloroacetic acid as an efficient and reusable catalytic system for the synthesis of new 1-((2-hydroxynaphthalen-1-yl) (phenyl) methyl) semicarbazides/thiosemicarbazides, Arabian J. Chem., 2017, 10, S730–S734. https://doi.org/10.1016/j.arabjc.2012.11.016.
- M.R. Maurya, B. Sarkar, A. Kumar, N. Ribeiro, A. Miliute, J.C. Pessoa, New thiosemicarbazide and dithiocarbazate based oxidovanadium(IV) and dioxidovanadium(V) complexes. Reactivity and catalytic potential, New J. Chem., 2019, 43(45), 17620–17635. https://doi.org/10.1039/C9NJ01486A.
- P.P. Netalkar, S.P. Netalkar, V.K. Revankar, Transition metal complexes of thiosemicarbazone: Synthesis, structures and invitro antimicrobial studies, Polyhedron, 2015, 100, 215–222. https://doi.org/10.1016/j.poly.2015.07.075.
- O.V. Dolomanov, L.J. Bourhis, R.J. Gildea, J.A.K. Howard, H. Puschmann, OLEX2: a complete structure solution, refinement and analysis program, J. Appl. Crystallogr., 2009, 42(2), 339–341. https://doi.org/10.1107/S0021889808042726.
- G.M. Sheldrick, SHELXT – Integrated space-group and crystal-structure determination, Acta Crystallogr., Sect. A: Found. Adv., 2015, C71, 3–8. https://doi.org/10.1107/S2053273314026370.
- G.M. Sheldrick, Crystal structure refinement with SHELXL, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71(1), 3–8. https://doi.org/10.1107/S2053229614024218.
- L.J. Farrugia, WinGX and ORTEP for Windows: an update, J. Appl. Crystallogr., 2012, 45(4), 849–854. https://doi.org/10.1107/S0021889812029111.
- J. Garcı́a-Tojal, A. Garcı́a-Orad, A.A. Dı́az, J.L. Serra, M.K. Urtiaga, M.I. Arriortua, T. Rojo, Biological activity of complexes derived from pyridine-2-carbaldehyde thiosemicarbazone: Structure of [Co(C7H7N4S)2][NCS], J. Inorg. Biochem., 2001, 84(3), 271–278. https://doi.org/10.1016/S0162-0134(01)00184-2.
- X. Fan, J. Dong, R. Min, Y. Chen, X. Yi, J. Zhou, S. Zhang, Cobalt(II) complexes with thiosemicarbazone as potential antitumor agents: synthesis, crystal structures, DNA interactions, and cytotoxicity, J. Coord. Chem., 2013, 66(24), 4268–4279. https://doi.org/10.1080/00958972.2013.867030.
- A. Baysal, M. Aydemir, F. Durap, S. Özkar, L.T. Yildirim, Synthesis and structural characterization of a novel seven-coordinate cobalt(II) complex: 2,9-Bis (ethanolamine)-1,10-phenanthrolinechlorocobalt(II) chloride, Inorg. Chim. Acta, 2011, 371(1), 107–110. https://doi.org/10.1016/j.ica.2011.03.050.
- P.P. Netalkar, S.P., Revankar, V.K. Netalkar, Transition metal complexes of thiosemicarbazone: Synthesis, structures and invitro antimicrobial studies, Polyhedron, 2015, 100, 215–222. https://doi.org/10.1016/j.poly.2015.07.075.
- A.V. Pestov, P.A. Slepukhin, O.V. Koryakova, V.N. Charushin, Nickel(II) and copper(II) complexes based on N-(2-carboxyethyl)alkanolamines: Influence of the amino alcohol structure on the coordination sphere of the metal center, Russian J. Coord. Chem., 2014, 40(4), 216–223. https://doi.org/10.1134/S107032841404006X.
- A.A. El-Sherif, A. Fetoh, Y.K. Abdulhamed, G.M.A. El-Reash, Synthesis, structural characterization, DFT studies and biological activity of Cu(II) and Ni(II) complexes of novel hydrazone, Inorg. Chim. Acta, 2018, 480, 1–15. https://doi.org/10.1016/j.ica.2018.04.038.
- A. Akbari, H. Ghateazadeh, R. Takjoo, B. Sadeghi-Nejad, M.Mehrvar, J.T. Mague, Synthesis crystal structures of four new biochemical active Ni(II) complexes of thiosemicarbazone and isothiosemicarbazone-based ligands: In vitro antimicrobial study, J. Mol. Struct., 2019, 1181,
–294. https://doi.org/10.1016/j.molstruc.2018.12.109.
- N.C. Saha, R.J. Butcher, S. Chaudhuri, N. Saha, Synthesis and spectroscopic characterisation of new nickel (II) complexes with 5-methyl-3-formylpyrazole-3-piperidinylthiosemicarbazone (HMPz3Pi): X-ray structures of HMPz3Pi and [Ni(HMPz3Pi)2]Cl2·2H2O with indication for unusual rotation about the azomethine double bond of the free ligand on complexation, Polyhedron, 2005, 24(9), 1015–1022. https://doi.org/10.1016/j.poly.2005.01.024.
- S.M. Kumar, K. Dhahagani, J. Rajesh, K. Nehru, J. Annaraj, G. Chakkaravarthi, G. Rajagopal, Synthesis, characterization, structural analysis and DNA binding studies of nickel(II)–triphenylphosphine complex of ONS donor ligand – Multisubstituted thiosemicarbazone as highly selective sensor for fluoride ion, Polyhedron, 2013, 59, 58–68. https://doi.org/10.1016/j.poly.2013.04.048.
- P. Anitha, R. Manikandan, P. Vijayan, G. Prakash, P. Viswanathamurthi, R.J. Butcher, Nickel(II) complexes containing ONS donor ligands: Synthesis, characterization, crystal structure and catalytic application towards C-C cross-coupling reactions, J. Chem. Sci., 2015, 127(4), 597–608. https://doi.org/10.1007/s12039-015-0811-4.
- L. Yang, D.R. Powell, R.P. Houser, Structural variation in copper (i) complexes with pyridylmethylamide ligands: structural analysis with a new four-coordinate geometry index, τ4, Dalton Trans., 2007, 955–964. https://doi.org/10.1039/B617136B.
- A.S. Abdelrazeq, H.A. Ghabbour, A.A. El-Emam, D.A. Osman, S. Garcia-Granda, Synthesis and crystal structure of 3-(adamantan-1-yl)-4-(2-bromo-4-fluorophenyl)-1H-1,2,4-triazole-5(4 H)-thione, Acta Crystallogr., Sect. E: Crystallogr. Commun., 2020, 76(2), 162–166. https://doi.org/10.1107/S2056989020000092.
- P.P. Netalkar, S.P. Netalkar, V.K. Revankar, Transition metal complexes of thiosemicarbazone: Synthesis, structures and invitro antimicrobial studies, Polyhedron, 2015, 100, 215–222. https://doi.org/10.1016/j.poly.2015.07.075.
- I.M. Shahid, M. Ahmad, M. Rahisuddin, R. Arif, S. Tasneem, F. Sama, I.A. Ansari, Synthesis, crystal structures, photoluminescence, magnetic and antioxidant properties, and theoretical analysis of Zn(II) and Cu(II) complexes of an aminoalcohol ligand supported by benzoate counter anions, New J. Chem., 2019, 43(2), 622–633. https://doi.org/10.1039/C8NJ04122A.
- M.K. Bharty, R.K. Dani, P. Nath, A. Bharti, N.K. Singh, O. Prakash, R.K. Singh, R.J. Butcher, Syntheses, structural and thermal studies on Zn(II) complexes of 5-aryl-1,3,4-oxadiazole-2-thione and dithiocarbamates: Antibacterial activity and DFT calculations, Polyhedron, 2015, 98, 84–95. https://doi.org/10.1016/j.poly.2015.05.045.
- B.S. Stadelman, J.M. Murphy, A.M. Owen, R. Castro-Ramírez, H.C. Smith, C.M. Cohen, L.X. Zhang, C.A. Bayse, C. D. McMillen, N. Barba-Behrens, J.L. Brumaghim, Zinc(II) thione and selone complexes: The effect of metal redox activity on ligand-based oxidation, Inorg. Chim. Acta, 2020, 502, 119379. https://doi.org/10.1016/j.ica.2019.119379.
- K. Karrouchi, S.A. Brandán, Y. Sert, M.E. Karbane, S. Radi, M. Ferbinteanu, Y, Garcia, M. Ansar, Synthesis, structural, molecular docking and spectroscopic studies of (E)-N’-(4-methoxybenzylidene)-5-methyl-1H-pyrazole-3- carbohydrazide, J. Mol. Chem., 2021, 1225, 129072. https://doi.org/10.1016/j.molstruc.2020.129072.
- C. Ndoye, G. Excoffier, G.A. Seck, O. Diouf, I.E. Thiam, M. Sidibe, M. Gaye, Crystal structures of bis[1-(1-hydroxypropan-2-ylidene)thiosemicarbazide-κ3S, N, O)cobalt(III)-tetra(thiocyanato-κN) cobalt(II) methanol solvate, bis{1-(1-hydroxypropan-2-ylidene) thiosemicarbazide-κ3S, N, O}nickel(II) bis(thiocyanate) and (1-(1-hydroxypropan-2-ylidene) thiosemicarbazide-κ3S, N, O)bis(thiocyanato-κN)zinc(II), Eur. J. Chem., 2022, 13(2), 196–205. https://doi.org/10.5155/eurjchem.13.2.196-205.2253.
- H. Sakiyama, K. Tone, M. Yamasaki, M. Mikuriya, Electronic spectrum and magnetic properties of a dinuclear nickel(II) complex with two nickel(II) ions of C2-twisted octahedral geometry, Inorg. Chim. Acta, 2011, 365(1), 183–189. https://doi.org/10.1016/j.ica.2010.09.005.
- Z. Trávníček, R. Pastorek, V. Slovák, Novel octahedral nickel(II) dithiocarbamates with bi- or tetradentate N-donor ligands: X-ray structures of [Ni(Bzppzdtc)(phen)2]ClO4·CHCl3 and [Ni(Bz2dtc)2(cyclam)], Polyhedron, 2008, 27(1), 411–419. https://doi.org/10.1016/j.poly.2007.09.024.
- K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds, (Fifth ed.), Wiley, New York, 1986.
- A.K. Srivastava, K. Srivastava, P. Yadav, J. Prasad, A.K. Maurya, Synthesis, characterization, biological (in vitro) activity and electrochemical studies of mixed-ligand copper (II) and cobalt (II) complexes with picolinic acid and imides, Chem. Data Collect., 2021, 31, 100620. https://doi.org/10.1016/j.cdc.2020.100620.
- M.H. Sadhu, A. Solanki, S.B. Kumar, Mixed ligand complexes of copper(II), cobalt(II), nickel(II) and zinc(II) with thiocyanate and pyrazole based tetradentate ligand: Syntheses, characterizations and structures, Polyhedron, 2015, 100, 206–214. https://doi.org/10.1016/j.poly.2015.07.067.
- N. Gupta, R. Gupta, S. Chandra, S.S. Bawa, Magnetic, electronic and electrochemical studies of mono and binuclear Cu(II) complexes using novel macrocyclic ligands, Spectrochim. Acta, Part A, 2005, 61(6), 1175–1180. https://doi.org/10.1016/j.saa.2004.06.038.
- W.J. Geary, The use of conductivity measurements in organic solvents for the characterisation of coordination compounds, Coord. Chem. Rev., 1971, 7(1), 81–122. https://doi.org/10.1016/S0010-8545(00)80009-0.
DOI: http://dx.doi.org/10.13171/mjc02305201688gaye
Refbacks
- There are currently no refbacks.
Copyright (c) 2023 Mediterranean Journal of Chemistry
