Kinetics of N-Deacetylation of Chitin Extracted from Shrimp Shells Collected from Coastal Area of Morocco
Abstract
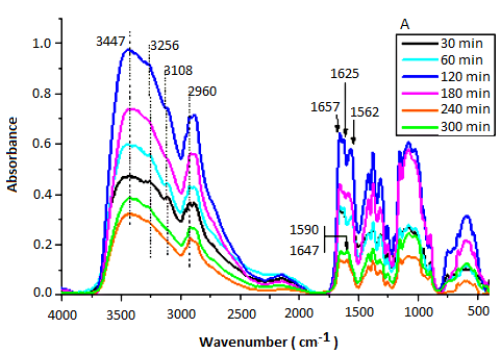
Chitosan was obtained from the alkaline N-deacetylation of ï¡-chitin, derived from shrimp shells (SS) collected from the Moroccan coast. Effects of temperature, NaOH concentration and reaction time on the kinetics of deacetylation were studied. The degree of deacetylation (DD = 75%) was obtained at T = 120 °C and CNaOH = 12N in a single step for 6h. It was found, from FTIR studies, that the removal of acetyl groups from chitin occur very fast for the reaction time t < 180 min and then becomes constant after, revealing two steps in the deacetylation of chitin following a first-order kinetics for each of them. The second step is considered the limiting step that has, in the temperature range of 25 – 120 °C and maintained CNaOH = 12N, a low apparent rate constant. The activation energy of this step is about 48.76 kJ/mol. The biopolymer (Chitin and chitosan) produced were characterized by X-ray diffraction.
Full Text:
PDFReferences
S. Bautista-Banõs, A.N. Hernández-Lauzardo, M.G. Velázquez-del Valle, M. Hernández- López, E. Ait Barka, E. Bosquez-Molina, C.L. Wilson, Crop Protection, 2006, 25, 108– 32 118.
Z. Cao, Z. H. Ge, S. Lai, European Polymer Journal, 2001, 37, 2141-2143.
M. Valentina Dinu, E. Stela Dragan, Chemical Engineering Journal, 2010, 160, 157–163.
M. Rinaudo, Progress in Polymer Science, 2006, 31, 603–632.
J. Iqbal, F.H. Wattoo, M.H. Sarwar Wattoo, R. Malik, S.A. Tirmizi, M. Imran, A.B. Ghangro, Arabian Journal of Chemistry, 2011, 4, 389–395.
M.R. Kasaai, Carbohydrate Polymers, 2008, 71, 497–508.
F.A. Al Sagheer, M.A. Al-Sughayer, S. Muslim, M.Z. Elsabee, Carbohydrate Polymers, 2009, 77, 410–419.
M.L. Duarte, M.C. Ferreira, M.R. Marvão, J. Rocha, International Journal of Biological Macromolecules, 2002, 31, 1- 8.
A. Tolaimate, J. Desbrières, M. Rhazi, A. Alagui, M. Vincendon, P. Vottero, Polymer, 2000, 41, 2463–2469.
P. Methacanon, M. Prasitsilp, T. Pothsree, J. Pattaraarchachai, Carbohydrate Polymers, 2003, 52, 119–123.
A. Chandumpai, N. Singhpibulporn, D. Faroongsarng, P. Sornprasit, Carbohydrate Polymers, 2004, 58, 467–474.
G. Galed, B. Miralles, I. Panõs, A. Santiago, Ã. Heras, Carbohydrate Polymers, 2005, 62, 316–320.
D. Ren, H. Yi, W. Wang, M. Xiaojun, Carbohydrate Research, 2005, 340, 2403–2410.
J. Brugnerotto, J. Lizardi, F.M. Goycoolea, W. ArguÈelles-Monal, J. Desbrie Ãres, M. Rinaudo, Polymer, 2001, 42, 3569 – 3580.
D. Baskar, T.S. Sampath Kumar, Carbohydrate Polymers, 2009, 78, 767–772.
W. Sajomsang, P. Gonil, Materials Science and Engineering C, 2010, 30, 357–363.
A. Percot, C. Viton, A. Domard, Biomacromolecules, 2003, 4, 1380-1385.
G. Chaussard, D. Domard, Biomacromolecules, 2004, 5, 559-564.
T.G. Liu, B. Li, W. Huang, B. Lv, J. Chen, J.X. Zhang, L.P. Zhu, Carbohydrate polymers, 2009, 77, 110–117.
R.L. Lavall, O.B.G. Assis, S.P. Campana-Filho, Bioresource Technology, 2007, 98, 2465–2472.
M. Fan, Q. Hu, K. Shen, Carbohydrate Polymers, 2009, 78, 66–71.
N. Yaghobi, F. Hormozi, Carbohydrate Polymers, 2010, 81, 892–896.
T. Lertwattanaseri, N. Ichikawa, T. Mizoguchi, Y. Tanaka, S. Chirachanchai, Carbohydrate Research, 2009, 344, 331–335.
Y. Zhang, C. Xue, Y. Xue, R. Gao, X. Zhang, Carbohydrate Research, 2005, 340, 1914–1917.
Y. Zhang, C. Xue, Z. Li, Y. Zhang, X. Fu, Carbohydrate Polymers, 2006, 65, 229–234.
K. Kurita, T. Sannan, Y. Iwakura, Makromolekulare Chemie, 1977, 178, 3197–3202.
T. Sannan, K. Kurita, Y. Iwakura, Polymer Journal, 1977, 9, 649-651.
A.A. Sarhan, D.M. Ayad, D.S. Badawy, M. Monier, Reactive & Functional Polymers, 2009, 69, 358.
DOI: http://dx.doi.org/10.13171/mjc.2.3.2013.22.01.20
Refbacks
- There are currently no refbacks.
Copyright (c) 2015 Mediterranean Journal of Chemistry
