Synthesis and crystal structure of tetra(quinolin-8-olato)thorium ethanol solvate, Th(C9H6NO)4*C2H5OH
Abstract
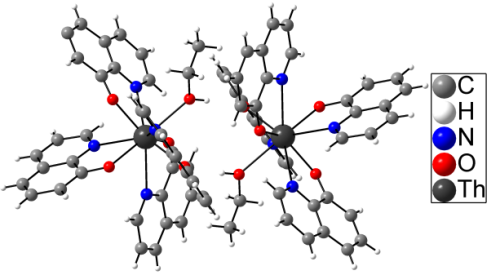
Metal quinolinolates (also known as oxinates) have been widely used for gravimetric analysis and in recent days as materials for organic light emitting diodes (OLEDs). Recrystallisation of tetra(quinolin-8-olato)thorium(IV) (ThQ4) yielded yellow, translucent, prismatic crystals of the title compound, ThQ4*C2H5OH, the single-crystal structure of which is described. Only one crystallographically distinct molecule is found, all ligands are crystallographically inequivalent. Thorium is coordinated by four 8-hydroxyquinolinato with average Th-O distances of 233.3 pm and average Th-N distances of 272.4 pm, and one ethanol ligand with a Th-O distance 256.7 pm. O and N surroundings around thorium can be described by a tricapped trigonal prism. In contrast to quinolinates of the trivalent metals, ThQ4*C2H5OH does not fluoresce under UV irradiation, which is in accordance with the lack of π-π overlap in the crystal structure.
Full Text:
PDFReferences
- M. Albrecht, M. Fiege, O. Osetska, Coord. Chem. Rev., 2008, 252, 812-824.
- M. Cölle, W. Brütting, Phys. Status Solidi A, 2004, 201, 1095-1115.
- N. M. Shavaleev, R. Scopelliti, F. Gumy, J.C.G. Bünzli, Inorg. Chem., 2009, 48, 7937-7946.
- C. Deraeve, M. Pitié, H. Mazarguil, B. Meunier, New J. Chem., 2007, 31, 193-195.
- M. A. Green, J. C. Hufman, J. Nucl. Med., 1988, 29, 417-420.
- G. M. da Costa, E. van San, E. De Grave, R. E. Vandenberghe, V. Barrón, L. Datas, Phys. Chem. Miner., 2002, 29, 122-131.
- J. H. Van Tassel, W. W. Wendtland, E. Sturm, J. Am. Chem. Soc., 1961, 83, 810-812.
- G. M. Sheldrick, SHELXL97 and SHELXS97, University of Göttingen, Germany, 1997.
- A. J. Freeman, C. Keller (Eds.), Handbook on the Physics and Chemistry of the Actinides, Vol. 4, North-Holland Physics-Publishing, Amsterdam, 1986. 10 - U. Benedict, J. Less-Common Met., 1987, 128, 7-45.
- H. Kohlmann, H. P. Beck, Z. Anorg. Allg. Chem., 1997, 623, 785-790. 12 - H. Kohlmann, H. P. Beck, Z. Kristallogr., 1999, 214, 341-345.
- Z. Szabó, T. Toraishi, V. Vallet, I Grenthe, Coord. Chem. Rev., 2006, 250, 784-815.
- R. K. Agarwal, P. C. Jain, V. Kapur, S Sharma, A. K. Srivastava, Transition Met. Chem. 1980, 5, 237-239.
- P. Thuery, Inorg. Chem. 2011, 50, 1898–1904.
- M. Brinkmann, G. Gadret, M. Muccini, C. Taliani, N. Masciocchi, A. Sironi, J. Am. Chem. Soc. 2000, 122, 5147-5157.
- L. S. Sapochak, A. Ranasinghe, H. Kohlmann, K. F. Ferris, P. E. Burrows, Chem. Mater., 2004, 16, 401-406.
- L. S. Sapochak, F. E. Benincasa, R. S. Schofield, J. L. Baker, K. K. C. Riccio, D.Fogarty, H. Kohlmann, K. F. Ferris, P. E. Burrows, J. Am. Chem. Soc., 2002, 124, 6119-6125.
DOI: http://dx.doi.org/10.13171/mjc.1.1.2011.27.05.13
Refbacks
- There are currently no refbacks.
Copyright (c) 2015 Mediterranean Journal of Chemistry
