Influence of the activation method on the activity of the nickel catalyst
Abstract
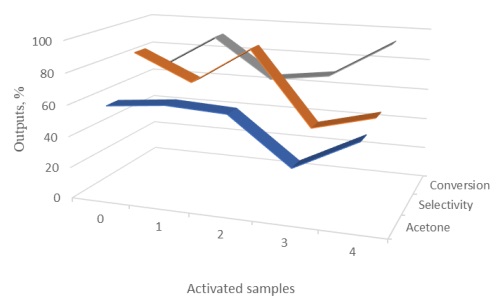
The results of a comparative analysis of the influence of the activation method on the catalytic properties of a nickel diatomaceous earth catalyst during the conversion of isopropyl alcohol to acetone are presented. Studies of the activity of the catalyst in the reaction of gas-phase dehydrogenation of isopropyl alcohol were carried out on a flow-type laboratory installation at a temperature of 150-350ºC. The mechanical activation of the nickel diatomaceous earth catalyst was carried out in a planetary mill. Traditional activation included pre-heat treatment of nickel diatomaceous earth catalyst in an atmosphere of O2 and H2 at 250°C; IR radiation was carried out using an incandescent lamp. It is shown that the nature of the activator determines the features of the formed surface nickel complexes. The activity of samples of the nickel diatomaceous earth catalyst subjected to activation by thermal reduction and thermal oxidation, as well as IR irradiation, testified to the low efficiency of these methods. A comparison of the activity of industrial nickel diatomaceous earth catalyst samples subjected to different activation methods showed that processing of samples by thermal reduction and thermal oxidation slightly increases the yield of acetone. In contrast, the original catalyst's IR irradiation and mechanical activation decreased its activity. Thus, thermal reduction and oxidation methods are promising for further research to increase the industrial nickel diatomaceous earth catalyst activity in dehydrogenating isopropyl alcohol into acetone.
Full Text:
PDFReferences
- S. Zafarnak, M.R. Rahimpour, Co-Ni bimetallic supported on mullite as a promising catalyst for dry biogas reforming toward hydrogen production, Molecular Catalysis, 2023, 534, 112803.
- D.-P. Phan, T.K. Vo, V.N. Le, J. Kim, E.Y. Lee, Spray pyrolysis synthesis of bimetallic NiMo/Al2O3–TiO2 catalyst for hydrodeoxygenation of guaiacol: Effects of bimetallic composition and reduction temperature, Journal of Industrial and Engineering Chemistry, 2020, 83, 351-358.
- A.V. Vutolkina., A.P. Glotov, A.V. Zanina, D.F. Makhmutov, A.L. Maximov, S.V. Egazar’yants, E.A. Karakhanov, Mesoporous Al-HMS, and Al-MCM-41 supported Ni-Mo sulfide catalysts for HYD and HDS via in situ hydrogen generation through a WGSR, Catalysis Today, 2019, 329, 156-166.
- H.G. El-Shobaky, Y.M. Fahmy, Nickel cuprate supported on cordierite as an active catalyst for CO oxidation by O2. Applied Catalysis B: Environmental, 2006, 63, 168-177.
- O.A. Knyazheva, O.N. Baklanova, A.V. Lavrenov, Mechanochemical synthesis of nanocrystalline nickel-molybdenum compounds, their morphology and application in catalysis: I. Effect of the Ni: Mo atomic ratio on the structure and properties of nickel-molybdenum compounds prepared under mechanochemical synthesis conditions, Kinetics and Catalysis, 2011, 52, 886-895.
- Z. Wang, L. Wang, Y. Cui, Y. Xing, W. Su, Research on nickel-based catalysts for carbon dioxide methanation combined with literature measurement, Journal of CO2 Utilization, 2022, 63, 102117.
- E.A. Guseinova, K.Y. Adzhamov, Electron properties of the system of Ni on kieselguhr. Russian Journal of Physical Chemistry A., 2011, 85, 1965-1970.
- E.A. Guseynova, K.Y. Adzhamov, Electronic properties of the nickel surface compound in the conversion of isopropyl alcohol, Herald of the Azerbaijan Engineering Academy, 2014, 6, 113-119.
- E. Guseynova, E. Zeynalov, K. Adzhamov, Physicochemical and catalytic properties of nano-nickel in converting isopropyl alcohol to acetone, Nanoindustry, 2014, 46-53.
- E.A. Guseinova, E.T. Zeinalov, K.Y. Adzhamov, Commercial nickel-kieselguhr isopropanol dehydrogenation catalysts: Morphology and catalytic and electronic properties, Catalysis in Industry, 2015, 7, 227 – 233.
- V.V. Zyryanov, Mechanochemical synthesis of complex oxides, Russian Chemical Reviews, 2008, 77, 105–135.
- V.V. Boldyrev, Mechanochemistry and mechanical activation of solids, Russian Chemical Reviews, 2006, 75, 177–189.
DOI: http://dx.doi.org/10.13171/mjc02305021696zeynalov
Refbacks
- There are currently no refbacks.
Copyright (c) 2023 Mediterranean Journal of Chemistry
